Lipid safety of tenofovir alafenamide during 96-week treatment in treatment-naive chronic hepatitis B patients
- PMID: 38895186
- PMCID: PMC11183333
- DOI: 10.3389/fmed.2024.1399665
Lipid safety of tenofovir alafenamide during 96-week treatment in treatment-naive chronic hepatitis B patients
Abstract
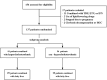
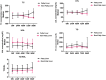
Background: This study was aimed at investigating the dynamics of lipids and the effect of TAF on the lipid profile of patients including fatty liver disease in CHB patients.
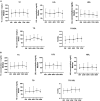
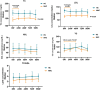
Methods: The data of TC, LDL-c, HDL-c, TG, and TC/HDL ratio were collected at baseline, 24 weeks, 48 weeks, 72 weeks, and 96 weeks. CHB patients with fatty liver at baseline were further analyzed in a subgroup.
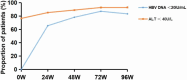
Results: A total of 137 CHB patients treated with TAF were enrolled in this study. During 96 weeks of TAF treatment, there was no significant change in TC, LDL-c, HDL-c, and TG level (P > 0.05). The TC/HDL-c ratio was increased with no significant change (+0.24, P > 0.05). In CHB patients with fatty liver (n = 48), TC, LDL-c, and TC/HDL-c ratio increased gradually during TAF treatment, TG levels increased to 146.63 mg/dL at 48 weeks (P = 0.057) and then decreased, but there was still no significant change compared with the baseline level by 96 weeks (P > 0.05).
Conclusion: TAF treatment had a low effect on the lipid profile of CHB patients over the course of 96 weeks, and it was safe even in patients with fatty liver.
Clinical trial registration: [https://www.chictr.org.cn/showproj.html?proj=65123], identifier [ChiCTR2000041005].
Keywords: 96-week; chronic hepatitis B; lipid safety; tenofovir alafenamide; treatment.
Copyright © 2024 Zhao, Liu, Zhang, Cui, Qu, Li, Wan, Wang, Chen, Shi, Li and Ye.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Comparison of lipid profile alterations in chronic hepatitis b patients receiving tenofovir alafenamide or tenofovir disoproxil fumarate.Sci Rep. 2024 Nov 9;14(1):27369. doi: 10.1038/s41598-024-78656-0. Sci Rep. 2024. PMID: 39521860 Free PMC article.
-
[Comparison of the effects of tenofovir amibufenamide and tenofovir alafenamide on lipid metabolism in the body].Zhonghua Gan Zang Bing Za Zhi. 2024 Dec 20;32(12):1123-1133. doi: 10.3760/cma.j.cn501113-20240901-00461. Zhonghua Gan Zang Bing Za Zhi. 2024. PMID: 39788587 Chinese.
-
Tenofovir alafenamide treatment may not worsen the lipid profile of chronic hepatitis B patients: A propensity score-matched analysis.Clin Mol Hepatol. 2022 Apr;28(2):254-264. doi: 10.3350/cmh.2021.0314. Epub 2021 Dec 28. Clin Mol Hepatol. 2022. PMID: 34959261 Free PMC article.
-
Risk of dyslipidemia in chronic hepatitis B patients taking tenofovir alafenamide: a systematic review and meta-analysis.Hepatol Int. 2023 Aug;17(4):860-869. doi: 10.1007/s12072-023-10528-7. Epub 2023 Apr 26. Hepatol Int. 2023. PMID: 37099248
-
Tenofovir alafenamide (TAF) treatment of HBV, what are the unanswered questions?Expert Rev Anti Infect Ther. 2018 Feb;16(2):153-161. doi: 10.1080/14787210.2018.1428561. Epub 2018 Jan 23. Expert Rev Anti Infect Ther. 2018. PMID: 29338458 Review.
Cited by
-
Metabolic effects and cardiovascular disease risks of TDF or TAF in patients with chronic hepatitis B: a systematic review and meta-analysis.Front Pharmacol. 2025 Jun 30;16:1604972. doi: 10.3389/fphar.2025.1604972. eCollection 2025. Front Pharmacol. 2025. PMID: 40661079 Free PMC article.
References
-
- European Association for the Study of the Liver. EASL 2017 clinical practice guidelines on the management of hepatitis B virus infection. J Hepatol. (2017) 67:370–98. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

