This is a preprint.
Glycolytic Disruption Triggers Interorgan Signaling to Nonautonomously Restrict Drosophila Larval Growth
- PMID: 38895259
- PMCID: PMC11185712
- DOI: 10.1101/2024.06.06.597835
Glycolytic Disruption Triggers Interorgan Signaling to Nonautonomously Restrict Drosophila Larval Growth
Abstract
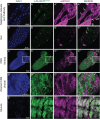
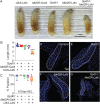
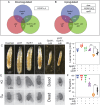
Drosophila larval growth requires efficient conversion of dietary nutrients into biomass. Lactate Dehydrogenase (Ldh) and Glycerol-3-phosphate dehydrogenase (Gpdh1) support larval biosynthetic metabolism by maintaining NAD+/NADH redox balance and promoting glycolytic flux. Consistent with the cooperative functions of Ldh and Gpdh1, the loss of both enzymes, but neither single enzyme, induces a developmental arrest. However, Ldh and Gpdh1 exhibit complex and often mutually exclusive expression patterns, suggesting that the Gpdh1; Ldh double mutant lethal phenotype could be mediated nonautonomously. Here we find that the developmental arrest displayed by the double mutants extends beyond simple metabolic disruption and instead stems, in part, from changes in systemic growth factor signaling. Specifically, we demonstrate that this synthetic lethality is linked to the upregulation of Upd3, a cytokine involved in the Jak/Stat signaling pathway. Moreover, we demonstrate that either loss of the Upd3 or dietary administration of the steroid hormone 20-hydroxyecdysone (20E) rescue the synthetic lethal phenotype of Gpdh1; Ldh double mutants. Together, these findings demonstrate that metabolic disruptions within a single tissue can nonautonomously modulate interorgan signaling to ensure synchronous developmental growth.
Keywords: Drosophila melanogaster; ecdysone; glycolysis; interorgan communication; metabolism.
Conflict of interest statement
COMPETING INTERESTS No competing interests declared
Figures

Similar articles
-
Glycolytic disruption restricts Drosophila melanogaster larval growth via the cytokine Upd3.PLoS Genet. 2025 May 2;21(5):e1011690. doi: 10.1371/journal.pgen.1011690. eCollection 2025 May. PLoS Genet. 2025. PMID: 40315265 Free PMC article.
-
Lactate dehydrogenase and glycerol-3-phosphate dehydrogenase cooperatively regulate growth and carbohydrate metabolism during Drosophila melanogaster larval development.Development. 2019 Sep 12;146(17):dev175315. doi: 10.1242/dev.175315. Development. 2019. PMID: 31399469 Free PMC article.
-
The Drosophila melanogaster enzyme glycerol-3-phosphate dehydrogenase 1 is required for oogenesis, embryonic development, and amino acid homeostasis.G3 (Bethesda). 2022 Jul 29;12(8):jkac115. doi: 10.1093/g3journal/jkac115. G3 (Bethesda). 2022. PMID: 35536221 Free PMC article.
-
Characterization of genetic and molecular tools for studying the endogenous expression of Lactate dehydrogenase in Drosophila melanogaster.PLoS One. 2024 Jan 3;19(1):e0287865. doi: 10.1371/journal.pone.0287865. eCollection 2024. PLoS One. 2024. PMID: 38170735 Free PMC article.
-
Ecdysone and 20-hydroxyecdysone are not required to activate glycolytic gene expression in Drosophila melanogaster embryos.MicroPubl Biol. 2021 Nov 30;2021:10.17912/micropub.biology.000501. doi: 10.17912/micropub.biology.000501. eCollection 2021. MicroPubl Biol. 2021. PMID: 34870111 Free PMC article.
References
-
- BAKER S. A. & RUTTER J. 2023. Metabolites as signalling molecules. Nat Rev Mol Cell Biol, 24, 355–374. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
