Spindle-locked ripples mediate memory reactivation during human NREM sleep
- PMID: 38898100
- PMCID: PMC11187142
- DOI: 10.1038/s41467-024-49572-8
Spindle-locked ripples mediate memory reactivation during human NREM sleep
Abstract
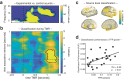
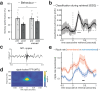
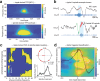
Memory consolidation relies in part on the reactivation of previous experiences during sleep. The precise interplay of sleep-related oscillations (slow oscillations, spindles and ripples) is thought to coordinate the information flow between relevant brain areas, with ripples mediating memory reactivation. However, in humans empirical evidence for a role of ripples in memory reactivation is lacking. Here, we investigated the relevance of sleep oscillations and specifically ripples for memory reactivation during human sleep using targeted memory reactivation. Intracranial electrophysiology in epilepsy patients and scalp EEG in healthy participants revealed that elevated levels of slow oscillation - spindle activity coincided with the read-out of experimentally induced memory reactivation. Importantly, spindle-locked ripples recorded intracranially from the medial temporal lobe were found to be correlated with the identification of memory reactivation during non-rapid eye movement sleep. Our findings establish ripples as key-oscillation for sleep-related memory reactivation in humans and emphasize the importance of the coordinated interplay of the cardinal sleep oscillations.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Endogenous memory reactivation during sleep in humans is clocked by slow oscillation-spindle complexes.Nat Commun. 2021 May 25;12(1):3112. doi: 10.1038/s41467-021-23520-2. Nat Commun. 2021. PMID: 34035303 Free PMC article.
-
Beyond spindles: interactions between sleep spindles and boundary frequencies during cued reactivation of motor memory representations.Sleep. 2018 Sep 1;41(9):zsy142. doi: 10.1093/sleep/zsy142. Sleep. 2018. PMID: 30137521 Free PMC article.
-
Sleep spindles mediate hippocampal-neocortical coupling during long-duration ripples.Elife. 2020 Jul 13;9:e57011. doi: 10.7554/eLife.57011. Elife. 2020. PMID: 32657268 Free PMC article.
-
Hijacking of hippocampal-cortical oscillatory coupling during sleep in temporal lobe epilepsy.Epilepsy Behav. 2021 Aug;121(Pt B):106608. doi: 10.1016/j.yebeh.2019.106608. Epub 2019 Nov 15. Epilepsy Behav. 2021. PMID: 31740330 Review.
-
Targeting sleep oscillations to improve memory in schizophrenia.Schizophr Res. 2020 Jul;221:63-70. doi: 10.1016/j.schres.2020.01.010. Epub 2020 Jan 31. Schizophr Res. 2020. PMID: 32014359 Free PMC article. Review.
Cited by
-
Efficacy and mechanisms of repeated closed-loop auditory exposure during slow-wave sleep for internet gaming disorder.Mol Psychiatry. 2025 Sep;30(9):4151-4160. doi: 10.1038/s41380-025-02995-1. Epub 2025 May 27. Mol Psychiatry. 2025. PMID: 40425853 Free PMC article. Clinical Trial.
-
Breathing orchestrates synchronization of sleep oscillations in the human hippocampus.Proc Natl Acad Sci U S A. 2024 Dec 24;121(52):e2405395121. doi: 10.1073/pnas.2405395121. Epub 2024 Dec 16. Proc Natl Acad Sci U S A. 2024. PMID: 39680758 Free PMC article.
-
A topological method of generating action potentials and electroencephalography oscillations in a surface network.R Soc Open Sci. 2025 May 28;12(5):241977. doi: 10.1098/rsos.241977. eCollection 2025 May. R Soc Open Sci. 2025. PMID: 40438545 Free PMC article.
-
Local inhibitory circuits mediate cortical reactivations and memory consolidation.Sci Adv. 2025 May 30;11(22):eadu9800. doi: 10.1126/sciadv.adu9800. Epub 2025 May 30. Sci Adv. 2025. PMID: 40446034 Free PMC article.
-
Electrophysiological signatures underlying variability in human memory consolidation.Nat Commun. 2025 Mar 12;16(1):2472. doi: 10.1038/s41467-025-57766-x. Nat Commun. 2025. PMID: 40074728 Free PMC article.
References
-
- Navarrete M, Valderrama M, Lewis PA. The role of slow-wave sleep rhythms in the cortical-hippocampal loop for memory consolidation. Curr. Opin. Behav. Sci. 2020;32:102–110. doi: 10.1016/j.cobeha.2020.02.006. - DOI
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

