Aging-induced short-chain acyl-CoA dehydrogenase promotes age-related hepatic steatosis by suppressing lipophagy
- PMID: 38898632
- PMCID: PMC11464120
- DOI: 10.1111/acel.14256
Aging-induced short-chain acyl-CoA dehydrogenase promotes age-related hepatic steatosis by suppressing lipophagy
Abstract
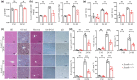
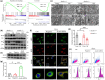
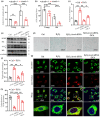
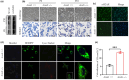
Hepatic steatosis, the first step in the development of nonalcoholic fatty liver disease (NAFLD), is frequently observed in the aging population. However, the underlying molecular mechanism remains largely unknown. In this study, we first employed GSEA enrichment analysis to identify short-chain acyl-CoA dehydrogenase (SCAD), which participates in the mitochondrial β-oxidation of fatty acids and may be associated with hepatic steatosis in elderly individuals. Subsequently, we examined SCAD expression and hepatic triglyceride content in various aged humans and mice and found that triglycerides were markedly increased and that SCAD was upregulated in aged livers. Our further evidence in SCAD-ablated mice suggested that SCAD deletion was able to slow liver aging and ameliorate aging-associated fatty liver. Examination of the molecular pathways by which the deletion of SCAD attenuates steatosis revealed that the autophagic degradation of lipid droplets, which was not detected in elderly wild-type mice, was maintained in SCAD-deficient old mice. This was due to the decrease in the production of acetyl-coenzyme A (acetyl-CoA), which is abundant in the livers of old wild-type mice. In conclusion, our findings demonstrate that the suppression of SCAD may prevent age-associated hepatic steatosis by promoting lipophagy and that SCAD could be a promising therapeutic target for liver aging and associated steatosis.
Keywords: acetyl‐coenzyme A; aging; autophagy; lipid droplet; lipophagy; liver steatosis; short‐chain acyl‐CoA dehydrogenase.
© 2024 The Author(s). Aging Cell published by Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Figures


Similar articles
-
Hepatocyte-secreted FAM3D ameliorates hepatic steatosis by activating FPR1-hnRNP U-GR-SCAD pathway to enhance lipid oxidation.Metabolism. 2023 Sep;146:155661. doi: 10.1016/j.metabol.2023.155661. Epub 2023 Jul 16. Metabolism. 2023. PMID: 37454871
-
Acetyl-CoA Derived from Hepatic Peroxisomal β-Oxidation Inhibits Autophagy and Promotes Steatosis via mTORC1 Activation.Mol Cell. 2020 Jul 2;79(1):30-42.e4. doi: 10.1016/j.molcel.2020.05.007. Epub 2020 May 29. Mol Cell. 2020. PMID: 32473093 Free PMC article.
-
Increased dietary fat contributes to dysregulation of the LKB1/AMPK pathway and increased damage in a mouse model of early-stage ethanol-mediated steatosis.J Nutr Biochem. 2013 Aug;24(8):1436-45. doi: 10.1016/j.jnutbio.2012.12.002. Epub 2013 Mar 1. J Nutr Biochem. 2013. PMID: 23465594 Free PMC article.
-
Molecular pathogenesis of a novel mutation, G108D, in short-chain acyl-CoA dehydrogenase identified in subjects with short-chain acyl-CoA dehydrogenase deficiency.Hum Genet. 2010 Jun;127(6):619-28. doi: 10.1007/s00439-010-0822-7. Epub 2010 Apr 8. Hum Genet. 2010. PMID: 20376488 Review.
-
The ménage à trois of autophagy, lipid droplets and liver disease.Autophagy. 2022 Jan;18(1):50-72. doi: 10.1080/15548627.2021.1895658. Epub 2021 Apr 2. Autophagy. 2022. PMID: 33794741 Free PMC article. Review.
References
-
- Ahrens, M. , Ammerpohl, O. , von Schonfels, W. , Kolarova, J. , Bens, S. , Itzel, T. , Teufel, A. , Herrmann, A. , Brosch, M. , Hinrichsen, H. , Erhart, W. , Egberts, J. , Sipos, B. , Schreiber, S. , Häsler, R. , Stickel, F. , Becker, T. , Krawczak, M. , Röcken, C. , … Hampe, J. (2013). DNA methylation analysis in nonalcoholic fatty liver disease suggests distinct disease‐specific and remodeling signatures after bariatric surgery. Cell Metabolism, 18, 296–302. - PubMed
-
- Carrer, A. , Parris, J. L. D. , Trefely, S. , Henry, R. A. , Montgomery, D. C. , Torres, A. , Viola, J. M. , Kuo, Y. M. , Blair, I. A. , Meier, J. L. , Andrews, A. J. , Snyder, N. W. , & Wellen, K. E. (2017). Impact of a high‐fat diet on tissue acyl‐CoA and histone acetylation levels. The Journal of Biological Chemistry, 292, 3312–3322. - PMC - PubMed
-
- Chalasani, N. , Younossi, Z. , Lavine, J. E. , Diehl, A. M. , Brunt, E. M. , Cusi, K. , Charlton, M. , & Sanyal, A. J. (2012). The diagnosis and management of non‐alcoholic fatty liver disease: Practice guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology, 55, 2005–2023. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

