Dosimetric risk factors for radiation esophagitis in patients with breast cancer following regional nodal radiation
- PMID: 38898857
- PMCID: PMC11185373
- DOI: 10.12998/wjcc.v12.i17.2995
Dosimetric risk factors for radiation esophagitis in patients with breast cancer following regional nodal radiation
Abstract
Background: Radiation esophagitis (RE) is one of the most common clinical symptoms of regi-onal lymph node radiotherapy for breast cancer. However, there are fewer studies focusing on RE caused by hypofractionated radiotherapy (HFRT).
Aim: To analyze the clinical and dosimetric factors that contribute to the development of RE in patients with breast cancer treated with HFRT of regional lymph nodes.
Methods: Between January and December 2022, we retrospectively analysed 64 patients with breast cancer who met our inclusion criteria underwent regional nodal intensity-modulated radiotherapy at a radiotherapy dose of 43.5 Gy/15F.
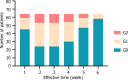
Results: Of the 64 patients in this study, 24 (37.5%) did not develop RE, 29 (45.3%) developed grade 1 RE (G1RE), 11 (17.2%) developed grade 2 RE (G2RE), and none developed grade 3 RE or higher. Our univariable logistic regression analysis found G2RE to be significantly correlated with the maximum dose, mean dose, relative volume 20-40, and absolute volume (AV) 20-40. Our stepwise linear regression analyses found AV30 and AV35 to be significantly associated with G2RE (P < 0.001). The optimal threshold for AV30 was 2.39 mL [area under the curve (AUC): 0.996; sensitivity: 90.9%; specificity: 91.1%]. The optimal threshold for AV35 was 0.71 mL (AUC: 0.932; sensitivity: 90.9%; specificity: 83.9%).
Conclusion: AV30 and AV35 were significantly associated with G2RE. The thresholds for AV30 and AV35 should be limited to 2.39 mL and 0.71 mL, respectively.
Keywords: Breast cancer; Dosimetric parameters; Esophagitis; Hypofractionated radiotherapy; Radiation esophagitis.
©The Author(s) 2024. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: Dr. Qi reports grants from the Handan City Science and Technology Research and Development Program.
Figures
Similar articles
-
Dose-Volume Predictors for Radiation Esophagitis in Patients With Breast Cancer Undergoing Hypofractionated Regional Nodal Radiation Therapy.Int J Radiat Oncol Biol Phys. 2023 Sep 1;117(1):186-197. doi: 10.1016/j.ijrobp.2023.03.060. Epub 2023 Mar 30. Int J Radiat Oncol Biol Phys. 2023. PMID: 37001764
-
Predictors of Acute Radiation Dermatitis and Esophagitis in African American Patients Receiving Whole-Breast Radiation Therapy.Pract Radiat Oncol. 2022 Jan-Feb;12(1):52-59. doi: 10.1016/j.prro.2021.08.004. Epub 2021 Oct 25. Pract Radiat Oncol. 2022. PMID: 34710629
-
[Analysis of risk factors of radiation-induced toxicity in limited-stage small cell lung cancer treated with hypofractionated intensity-modulated radiotherapy].Zhonghua Zhong Liu Za Zhi. 2023 Jul 23;45(7):627-633. doi: 10.3760/cma.j.cn112152-20221031-00733. Zhonghua Zhong Liu Za Zhi. 2023. PMID: 37462020 Chinese.
-
Dosimetric parameters associated with radiation-induced esophagitis in breast cancer patients undergoing regional nodal irradiation.Radiother Oncol. 2021 Feb;155:167-173. doi: 10.1016/j.radonc.2020.10.042. Epub 2020 Nov 4. Radiother Oncol. 2021. PMID: 33157173
-
Hypofractionated radiotherapy after breast-conserving surgery: Clinical and dosimetric factors predictive of acute skin toxicity.Strahlenther Onkol. 2023 Jan;199(1):48-54. doi: 10.1007/s00066-022-01985-4. Epub 2022 Aug 9. Strahlenther Onkol. 2023. PMID: 35943552
References
-
- Ragaz J, Jackson SM, Le N, Plenderleith IH, Spinelli JJ, Basco VE, Wilson KS, Knowling MA, Coppin CM, Paradis M, Coldman AJ, Olivotto IA. Adjuvant radiotherapy and chemotherapy in node-positive premenopausal women with breast cancer. N Engl J Med. 1997;337:956–962. - PubMed
-
- Krug D, Baumann R, Combs SE, Duma MN, Dunst J, Feyer P, Fietkau R, Haase W, Harms W, Hehr T, Piroth MD, Sedlmayer F, Souchon R, Strnad V, Budach W Breast Cancer Expert Panel of the German Society of Radiation Oncology (DEGRO) Moderate hypofractionation remains the standard of care for whole-breast radiotherapy in breast cancer: Considerations regarding FAST and FAST-Forward. Strahlenther Onkol. 2021;197:269–280. - PMC - PubMed
-
- Wang SL, Fang H, Song YW, Wang WH, Hu C, Liu YP, Jin J, Liu XF, Yu ZH, Ren H, Li N, Lu NN, Tang Y, Qi SN, Sun GY, Peng R, Li S, Chen B, Yang Y, Li YX. Hypofractionated versus conventional fractionated postmastectomy radiotherapy for patients with high-risk breast cancer: a randomised, non-inferiority, open-label, phase 3 trial. Lancet Oncol. 2019;20:352–360. - PubMed
-
- START Trialists' Group; Bentzen SM, Agrawal RK, Aird EG, Barrett JM, Barrett-Lee PJ, Bentzen SM, Bliss JM, Brown J, Dewar JA, Dobbs HJ, Haviland JS, Hoskin PJ, Hopwood P, Lawton PA, Magee BJ, Mills J, Morgan DA, Owen JR, Simmons S, Sumo G, Sydenham MA, Venables K, Yarnold JR. The UK Standardisation of Breast Radiotherapy (START) Trial B of radiotherapy hypofractionation for treatment of early breast cancer: a randomised trial. Lancet. 2008;371:1098–1107. - PMC - PubMed
-
- Murray Brunt A, Haviland JS, Wheatley DA, Sydenham MA, Alhasso A, Bloomfield DJ, Chan C, Churn M, Cleator S, Coles CE, Goodman A, Harnett A, Hopwood P, Kirby AM, Kirwan CC, Morris C, Nabi Z, Sawyer E, Somaiah N, Stones L, Syndikus I, Bliss JM, Yarnold JR FAST-Forward Trial Management Group. Hypofractionated breast radiotherapy for 1 week versus 3 weeks (FAST-Forward): 5-year efficacy and late normal tissue effects results from a multicentre, non-inferiority, randomised, phase 3 trial. Lancet. 2020;395:1613–1626. - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

