Blebbistatin as a novel antiviral agent targeting equid herpesvirus type 8
- PMID: 38898998
- PMCID: PMC11186319
- DOI: 10.3389/fvets.2024.1390304
Blebbistatin as a novel antiviral agent targeting equid herpesvirus type 8
Abstract
Introduction: Equid herpesvirus type 8 (EqHV-8) poses a significant threat to equine health, leading to miscarriages and respiratory diseases in horses and donkeys, and results in substantial economic losses in the donkey industry. Currently, there are no effective drugs or vaccines available for EqHV-8 infection control.
Methods: In this study, we investigated the in vitro and in vivo antiviral efficacy of Blebbistatin, a myosin II ATPase inhibitor, against EqHV-8.
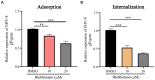
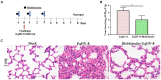
Results: Our results demonstrated that Blebbistatin significantly inhibited EqHV-8 infection in Rabbit kidney (RK-13) and Madin-Darby Bovine Kidney (MDBK) cells in a concentration-dependent manner. Notably, Blebbistatin was found to disrupt EqHV-8 infection at the entry stage by modulating myosin II ATPase activity. Moreover, in vivo experiments revealed that Blebbistatin effectively reduced EqHV-8 replication and mitigated lung pathology in a mouse model.
Conclusion: Collectively, these findings suggest that Blebbistatin holds considerable potential as an antiviral agent for the control of EqHV-8 infection, presenting a novel approach to addressing this veterinary challenge.
Keywords: EqHV-8; animal model; antiviral activity; blebbistatin; myosin II ATPase inhibitor.
Copyright © 2024 Li, Cui, Yu, Sun, Li, Li, Li, Chen, Khan, Wang and Wang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Protective Role of Cepharanthine Against Equid Herpesvirus Type 8 Through AMPK and Nrf2/HO-1 Pathway Activation.Viruses. 2024 Nov 12;16(11):1765. doi: 10.3390/v16111765. Viruses. 2024. PMID: 39599879 Free PMC article.
-
Rutin prevents EqHV-8 induced infection and oxidative stress via Nrf2/HO-1 signaling pathway.Front Cell Infect Microbiol. 2024 Apr 25;14:1386462. doi: 10.3389/fcimb.2024.1386462. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 38725448 Free PMC article.
-
Cobalt Protoporphyrin Blocks EqHV-8 Infection via IFN-α/β Production.Animals (Basel). 2023 Aug 22;13(17):2690. doi: 10.3390/ani13172690. Animals (Basel). 2023. PMID: 37684954 Free PMC article.
-
Antiviral agents against equid alphaherpesviruses: Current status and perspectives.Vet J. 2016 Jan;207:38-44. doi: 10.1016/j.tvjl.2015.06.010. Epub 2015 Jun 12. Vet J. 2016. PMID: 26654843 Review.
-
Medicinal Chemistry and Use of Myosin II Inhibitor ( S)-Blebbistatin and Its Derivatives.J Med Chem. 2018 Nov 8;61(21):9410-9428. doi: 10.1021/acs.jmedchem.8b00503. Epub 2018 Jun 21. J Med Chem. 2018. PMID: 29878759 Review.
Cited by
-
Evaluation of Celastrol Antiviral Activity Against Equid Alphaherpesvirus Type 8 Infection.Viruses. 2025 Feb 28;17(3):347. doi: 10.3390/v17030347. Viruses. 2025. PMID: 40143276 Free PMC article.
-
Equine Herpesvirus-1 Induced Respiratory Disease in Dezhou Donkey Foals: Case Study from China, 2024.Vet Sci. 2025 Jan 14;12(1):56. doi: 10.3390/vetsci12010056. Vet Sci. 2025. PMID: 39852931 Free PMC article.
-
Advances in Donkey Disease Surveillance and Microbiome Characterization in China.Microorganisms. 2025 Mar 26;13(4):749. doi: 10.3390/microorganisms13040749. Microorganisms. 2025. PMID: 40284586 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

