Cytotoxicity of phosphoramidate, bis-amidate and cycloSal prodrug metabolites against tumour and normal cells
- PMID: 38903945
- PMCID: PMC11109934
- DOI: 10.1039/d4md00115j
Cytotoxicity of phosphoramidate, bis-amidate and cycloSal prodrug metabolites against tumour and normal cells
Abstract
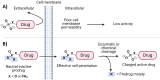
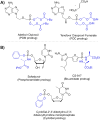
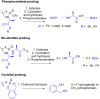
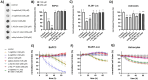
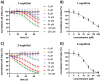
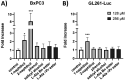
Phosphonate and phosphate prodrugs are integral to enhancing drug permeability, but the potential toxicity of their metabolites requires careful consideration. This study evaluates the impact of widely used phosphoramidate, bis-amidate, and cycloSal phosph(on)ate prodrug metabolites on BxPC3 pancreatic cancer cells, GL261-Luc glioblastoma cells, and primary cultured mouse astrocytes. 1-Naphthol and 2-naphthol demonstrated the greatest toxicity. Notably, 2-naphthol exhibited an ED50 of 21 μM on BxPC3 cells, surpassing 1-naphthol with an ED50 of 82 μM. Real-time xCELLigence experiments revealed notable activity for both metabolites at a low concentration of 16 μM. On primary cultured mouse astrocyte cells, all prodrugs exhibited reduced viability at 128 to 256 μM after only 4 hours of exposure. A cell-type-dependent sensitivity to phosph(on)ate prodrug metabolites was evident, with normal cells showing greater susceptibility than corresponding tumour cells. The results suggest it is essential to consider the potential cytotoxicity of phosph(on)ate prodrugs in the drug design and evaluation process.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There is no conflict of interest to declare.
Figures
Similar articles
-
Efficiency of bis-amidate phosphonate prodrugs.Bioorg Med Chem Lett. 2022 Jun 15;66:128724. doi: 10.1016/j.bmcl.2022.128724. Epub 2022 Apr 8. Bioorg Med Chem Lett. 2022. PMID: 35405283 Free PMC article.
-
Comparative Pharmacology of a Bis-Pivaloyloxymethyl Phosphonate Prodrug Inhibitor of Enolase after Oral and Parenteral Administration.ACS Pharmacol Transl Sci. 2023 Jan 6;6(2):245-252. doi: 10.1021/acsptsci.2c00216. eCollection 2023 Feb 10. ACS Pharmacol Transl Sci. 2023. PMID: 36798479 Free PMC article.
-
Design, synthesis, and characterization of a series of cytochrome P(450) 3A-activated prodrugs (HepDirect prodrugs) useful for targeting phosph(on)ate-based drugs to the liver.J Am Chem Soc. 2004 Apr 28;126(16):5154-63. doi: 10.1021/ja031818y. J Am Chem Soc. 2004. PMID: 15099098
-
Novel approaches for designing 5'-O-ester prodrugs of 3'-azido-2', 3'-dideoxythymidine (AZT).Curr Med Chem. 2000 Oct;7(10):995-1039. doi: 10.2174/0929867003374372. Curr Med Chem. 2000. PMID: 10911016 Review.
-
Application of the cycloSal-prodrug approach for improving the biological potential of phosphorylated biomolecules.Antiviral Res. 2006 Sep;71(2-3):282-92. doi: 10.1016/j.antiviral.2006.04.011. Epub 2006 May 9. Antiviral Res. 2006. PMID: 16735066 Review.
Cited by
-
The key phosphorus moieties in drug design: antimicrobials and beyond.Future Med Chem. 2024;16(23):2455-2458. doi: 10.1080/17568919.2024.2423602. Epub 2024 Nov 19. Future Med Chem. 2024. PMID: 39560019 No abstract available.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

