Anti-acetylated-tau immunotherapy is neuroprotective in tauopathy and brain injury
- PMID: 38915105
- PMCID: PMC11197196
- DOI: 10.1186/s13024-024-00733-9
Anti-acetylated-tau immunotherapy is neuroprotective in tauopathy and brain injury
Abstract
Background: Tau is aberrantly acetylated in various neurodegenerative conditions, including Alzheimer's disease, frontotemporal lobar degeneration (FTLD), and traumatic brain injury (TBI). Previously, we reported that reducing acetylated tau by pharmacologically inhibiting p300-mediated tau acetylation at lysine 174 reduces tau pathology and improves cognitive function in animal models.
Methods: We investigated the therapeutic efficacy of two different antibodies that specifically target acetylated lysine 174 on tau (ac-tauK174). We treated PS19 mice, which harbor the P301S tauopathy mutation that causes FTLD, with anti-ac-tauK174 and measured effects on tau pathology, neurodegeneration, and neurobehavioral outcomes. Furthermore, PS19 mice received treatment post-TBI to evaluate the ability of the immunotherapy to prevent TBI-induced exacerbation of tauopathy phenotypes. Ac-tauK174 measurements in human plasma following TBI were also collected to establish a link between trauma and acetylated tau levels, and single nuclei RNA-sequencing of post-TBI brain tissues from treated mice provided insights into the molecular mechanisms underlying the observed treatment effects.
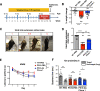
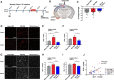
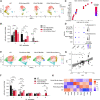
Results: Anti-ac-tauK174 treatment mitigates neurobehavioral impairment and reduces tau pathology in PS19 mice. Ac-tauK174 increases significantly in human plasma 24 h after TBI, and anti-ac-tauK174 treatment of PS19 mice blocked TBI-induced neurodegeneration and preserved memory functions. Anti-ac-tauK174 treatment rescues alterations of microglial and oligodendrocyte transcriptomic states following TBI in PS19 mice.
Conclusions: The ability of anti-ac-tauK174 treatment to rescue neurobehavioral impairment, reduce tau pathology, and rescue glial responses demonstrates that targeting tau acetylation at K174 is a promising neuroprotective therapeutic approach to human tauopathies resulting from TBI or genetic disease.
Keywords: Acetylated tau; Human plasma; Immunotherapy; TBI; Tauopathy.
© 2024. The Author(s).
Conflict of interest statement
LG is founder and equity holder of Aeton Therapeutics Inc.
Figures



References
MeSH terms
Substances
Grants and funding
- P30 AG062428/AG/NIA NIH HHS/United States
- R01 AG056770/AG/NIA NIH HHS/United States
- R01 AG074273/AG/NIA NIH HHS/United States
- The Valour Foundation/The Valour Foundation
- R25GM130494/GM/NIGMS NIH HHS/United States
- U01 AG073323/AG/NIA NIH HHS/United States
- R01 AG066707/AG/NIA NIH HHS/United States
- R01 AG072758/AG/NIA NIH HHS/United States
- RM1GM142002/GM/NIGMS NIH HHS/United States
- W81XWH-22-1-0129/U.S. Department of Defense
- R01AG071512/AG/NIA NIH HHS/United States
- R01AG056770/AG/NIA NIH HHS/United States
- K99 AG053439/AG/NIA NIH HHS/United States
- Rogers Family Foundation/Rogers Family Foundation
- Louis Stokes VA Medical Center/Louis Stokes VA Medical Center
- U54NS100717/NS/NINDS NIH HHS/United States
- R01 AG071512/AG/NIA NIH HHS/United States
- Tau Consortium/Tau Consortium
- U54 NS100717/NS/NINDS NIH HHS/United States
- R01AG054214/AG/NIA NIH HHS/United States
- I01BX005976/U.S. Department of Veterans Affairs
- T32AG071474/AG/NIA NIH HHS/United States
- U01AG073323/AG/NIA NIH HHS/United States
- JPB Foundation/JPB Foundation
- R01AG072758/AG/NIA NIH HHS/United States
- R01 AG074541/AG/NIA NIH HHS/United States
- RM1 GM142002/GM/NIGMS NIH HHS/United States
- R01 AG078185/AG/NIA NIH HHS/United States
- R01AG074273/AG/NIA NIH HHS/United States
- T32 AG071474/AG/NIA NIH HHS/United States
- R01AG066707/AG/NIA NIH HHS/United States
- R01 AG054214/AG/NIA NIH HHS/United States
- 19PABH13458000/American Heart Association
- R01AG074541/AG/NIA NIH HHS/United States
- R01AG078185/AG/NIA NIH HHS/United States
- P30AG062428/AG/NIA NIH HHS/United States
- R25 GM130494/GM/NIGMS NIH HHS/United States
- I01 BX005976/BX/BLRD VA/United States
- T32GM007250/GM/NIGMS NIH HHS/United States
- P30 AG072959/AG/NIA NIH HHS/United States
- T32 GM007250/GM/NIGMS NIH HHS/United States
- G.R. Lincoln Family Foundation/G.R. Lincoln Family Foundation
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

