Biomarkers in clinical epidemiology studies
- PMID: 38915440
- PMCID: PMC11195614
- DOI: 10.1093/ckj/sfae130
Biomarkers in clinical epidemiology studies
Erratum in
-
Correction to: Biomarkers in clinical epidemiology studies.Clin Kidney J. 2024 Sep 28;17(9):sfae293. doi: 10.1093/ckj/sfae293. eCollection 2024 Sep. Clin Kidney J. 2024. PMID: 39345687 Free PMC article.
Abstract
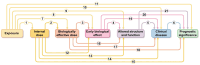
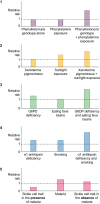
This paper discusses the use of biomarkers in clinical practice and biomedical research. Biomarkers are measurable characteristics that can be used to indicate the presence or absence of a disease or to track the progression of a disease. They can also be used to predict how a patient will respond to a particular treatment. Biomarkers have enriched clinical practice and disease prognosis by providing measurable characteristics that indicate biological processes. They offer valuable insights into disease susceptibility, progression, and treatment response, aiding drug development and personalized medicine. However, developing and implementing biomarkers come with challenges that must be addressed. Rigorous testing, standardization of assays, and consideration of ethical factors are crucial in ensuring the reliability and validity of biomarkers. Reliability is vital in biomarker research. It ensures accurate measurements by preventing biases and facilitating robust correlations with outcomes. Conversely, validation examines which and how many biomarkers correspond to theoretical constructs and external criteria, establishing their predictive value. Multiple biomarkers are sometimes necessary to represent the complex relationship between exposure and disease outcomes accurately. Susceptibility factors are pivotal in disease states' complex interaction among genetic and environmental factors. Gaining a comprehensive understanding of these factors is essential for effectively interpreting biomarker data and maximizing their clinical usefulness. Using well-validated biomarkers can improve diagnoses, more effective treatment evaluations, and enhanced disease prediction. This, in turn, will contribute to better patient outcomes and drive progress in medicine.
Keywords: CKD; biomarkers; cardiovascular; epidemiology; inflammation.
© The Author(s) 2024. Published by Oxford University Press on behalf of the ERA.
Conflict of interest statement
C.Z. is member of the Clinical Kidney Journal Editorial Board.
Figures
Comment in
-
Correction to: Biomarkers in clinical epidemiology studies.Clin Kidney J. 2024 Sep 28;17(9):sfae293. doi: 10.1093/ckj/sfae293. eCollection 2024 Sep. Clin Kidney J. 2024. PMID: 39345687 Free PMC article.
References
-
- Silver Spring (MD): Food and Drug Administration (US); Bethesda (MD): National Institutes of Health (US): BEST (Biomarkers, EndpointS, and other Tools) Resource . BEST (Biomarkers, EndpointS, and other Tools) Resource. https://www.ncbi.nlm.nih.gov/books/NBK326791/2021; (April 2023, date last accessed).
-
- Hulka BS, Wilcosky T. Biological markers in epidemiologic research. Arch Environ Health 1988;43:83–89. http://www.tandfonline.com/doi/abs/10.1080/00039896.1988.9935831 - DOI - PubMed
-
- Schulte PA. A conceptual framework for the validation and use of biologic markers. Environ Res 1989;48:129–44. https://linkinghub.elsevier.com/retrieve/pii/S0013935189800295 - PubMed
Publication types
LinkOut - more resources
Full Text Sources

