The Effect of Exposure to SARS-CoV-2 Vaccination and Infection on Humoral and Cellular Immunity in a Cohort of Patients with Immune-Mediated Diseases: A Pilot Study
- PMID: 38921803
- PMCID: PMC11206661
- DOI: 10.3390/pathogens13060506
The Effect of Exposure to SARS-CoV-2 Vaccination and Infection on Humoral and Cellular Immunity in a Cohort of Patients with Immune-Mediated Diseases: A Pilot Study
Abstract
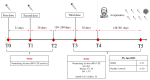
Immunization against COVID-19 is needed in patients with immune-mediated inflammatory diseases (IMIDs). However, data on long-term immunity kinetics remain scarce. This study aimed to compare the humoral and cellular response to COVID-19 in patients with immune-mediated inflammatory diseases (IMIDs) compared to healthy controls. We compared the humoral and cellular response to SARS-Cov-2 elicited by vaccination and/or infection in a prospective cohort of 20 IMID patients compared with a group of 21 healthcare workers (HCWs). We assessed immunity before and after the third and fourth dose of BNT162b2 or after COVID-19 infection using quantitative IgG anti-SARS-CoV-2 Spike antibody (anti-S-IgG), neutralization assay, and specific interferon-gamma (IFN-g) release assay (IGRA). The responses were compared with those of healthy controls. The two groups were similar in age and total exposure, becoming infected for the first time, mainly after the third dose. Neutralizing antibodies and IGRA were negative in 9.5% of IMID patients but not in any HCWs. No significant difference was found between neutralization titers to BA.1 in the IMID and the HCW groups. The study highlights the SARS-CoV-2 immunological responses in healthy controls and IMID patients, suggesting that the combined stimuli of vaccination and infection in IMID patients could promote a more profound immunological response.
Keywords: COVID–19; immune-mediated diseases; infections; interferon-gamma release assay; vaccine.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Evaluation of humoral and cellular immune responses in healthcare workers with varying levels of SARS-CoV-2 exposure: effects of CoronaVac vaccination followed by heterologous booster.Front Immunol. 2025 May 8;16:1576430. doi: 10.3389/fimmu.2025.1576430. eCollection 2025. Front Immunol. 2025. PMID: 40406109 Free PMC article.
-
Heterologous and homologous COVID-19 mRNA vaccination schemes for induction of basic immunity show similar immunogenicity regarding long-term spike-specific cellular immunity in healthcare workers.Vaccine. 2024 Aug 30;42(21):126132. doi: 10.1016/j.vaccine.2024.07.033. Epub 2024 Jul 20. Vaccine. 2024. PMID: 39034219
-
Humoral and cellular immune durability of different COVID-19 vaccine platforms following homologous/heterologous boosters: one-year post vaccination.Front Immunol. 2025 Jan 22;16:1526444. doi: 10.3389/fimmu.2025.1526444. eCollection 2025. Front Immunol. 2025. PMID: 39911379 Free PMC article.
-
Humoral immunity and safety of respiratory virus vaccines in systemic lupus erythematosus population: a meta-analysis based on twenty-five observational studies.Ann Med. 2024 Dec;56(1):2392882. doi: 10.1080/07853890.2024.2392882. Epub 2024 Aug 19. Ann Med. 2024. PMID: 39155852 Free PMC article.
-
Mechanisms and implications of IgG4 responses to SARS-CoV-2 and other repeatedly administered vaccines.J Infect. 2024 Dec;89(6):106317. doi: 10.1016/j.jinf.2024.106317. Epub 2024 Oct 16. J Infect. 2024. PMID: 39419185 Review.
Cited by
-
Did We Overreact? Insights on COVID-19 Disease and Vaccination in a Large Cohort of Immune-Mediated Inflammatory Disease Patients during Sequential Phases of the Pandemic (The BELCOMID Study).Vaccines (Basel). 2024 Oct 11;12(10):1157. doi: 10.3390/vaccines12101157. Vaccines (Basel). 2024. PMID: 39460324 Free PMC article.
References
-
- Goel R.R., Painter M.M., Apostolidis S.A., Mathew D., Meng W., Rosenfeld A.M., Lundgreen K.A., Reynaldi A., Khoury D.S., Pattekar A., et al. mRNA vaccines induce durable immune memory to SARS-CoV-2 and variants of concern. Science. 2021;374:abm0829. doi: 10.1126/science.abm0829. - DOI - PMC - PubMed
-
- Williams J.P., Meyers J.A. Immune-mediated inflammatory disorders (I.M.I.D.s): The economic and clinical costs. Am. J. Manag. Care. 2002;8:S664–S681. quiz S682–S685. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

