Accelerated evolution in the human lineage led to gain and loss of transcriptional enhancers in the RBFOX1 locus
- PMID: 38924416
- PMCID: PMC11204294
- DOI: 10.1126/sciadv.adl1049
Accelerated evolution in the human lineage led to gain and loss of transcriptional enhancers in the RBFOX1 locus
Abstract
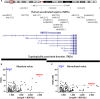
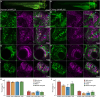
A long-standing goal of evolutionary biology is to decode how changes in gene regulatory networks contribute to human-specific traits. Human accelerated regions (HARs) are prime candidates for driving gene regulatory modifications in human development. The RBFOX1 locus is densely populated with HARs, providing a set of potential regulatory elements that could have changed its expression in the human lineage. Here, we examined the role of RBFOX1-HARs using transgenic zebrafish reporter assays and identified 15 transcriptional enhancers that are active in the developing nervous system, 9 of which displayed differential activity between the human and chimpanzee sequences. The engineered loss of two selected RBFOX1-HARs in knockout mouse models modified Rbfox1 expression at specific developmental stages and tissues in the brain, influencing the expression and splicing of a high number of Rbfox1 target genes. Our results provided insight into the spatial and temporal changes in gene expression driven by RBFOX1-HARs.
Figures




References
-
- King M. C., Wilson A. C., Evolution at two levels in humans and chimpanzees. Science 188, 107–116 (1975). - PubMed
-
- Pollard K. S., Salama S. R., Lambert N., Lambot M.-A., Coppens S., Pedersen J. S., Katzman S., King B., Onodera C., Siepel A., Kern A. D., Dehay C., Igel H., Ares M. Jr., Vanderhaeghen P., Haussler D., An RNA gene expressed during cortical development evolved rapidly in humans. Nature 443, 167–172 (2006). - PubMed
-
- Lindblad-Toh K., Garber M., Zuk O., Lin M. F., Parker B. J., Washietl S., Kheradpour P., Ernst J., Jordan G., Mauceli E., Ward L. D., Lowe C. B., Holloway A. K., Clamp M., Gnerre S., Alföldi J., Beal K., Chang J., Clawson H., Cuff J., Palma F. D., Fitzgerald S., Flicek P., Guttman M., Hubisz M. J., Jaffe D. B., Jungreis I., Kent W. J., Kostka D., Lara M., Martins A. L., Massingham T., Moltke I., Raney B. J., Rasmussen M. D., Robinson J., Stark A., Vilella A. J., Wen J., Xie X., Zody M. C.; Broad Institute Sequencing Platform and Whole Genome Assembly Team, Baldwin J., Bloom T., Chin C. W., Heiman D., Nicol R., Nusbaum C., Young S., Wilkinson J., Worley K. C., Kovar C. L., Muzny D. M., Gibbs R. A.; Baylor College of Medicine Human Genome Sequencing Center Sequencing Team, Cree A., Dihn H. H., Fowler G., Jhangiani S., Joshi V., Lee S., Lewis L. R., Nazareth L. V., Okwuonu G., Santibanez J., Warren W. C., Mardis E. R., Weinstock G. M., Wilson R. K.; Genome Institute at Washington University, Delehaunty K., Dooling D., Fronik C., Fulton L., Fulton B., Graves T., Minx P., Sodergren E., Birney E., Margulies E. H., Herrero J., Green E. D., Haussler D., Siepel A., Goldman N., Pollard K. S., Pedersen J. S., Lander E. S., Kellis M., A high-resolution map of human evolutionary constraint using 29 mammals. Nature 478, 476–482 (2011). - PMC - PubMed
-
- Prabhakar S., Noonan J. P., Pääbo S., Rubin E. M., Accelerated evolution of conserved noncoding sequences in humans. Science 314, 786 (2006). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

