Dietary Supplementation with Naringin Improves Systemic Metabolic Status and Alleviates Oxidative Stress in Transition Cows via Modulating Adipose Tissue Function: A Lipid Perspective
- PMID: 38929076
- PMCID: PMC11200899
- DOI: 10.3390/antiox13060638
Dietary Supplementation with Naringin Improves Systemic Metabolic Status and Alleviates Oxidative Stress in Transition Cows via Modulating Adipose Tissue Function: A Lipid Perspective
Abstract
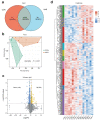
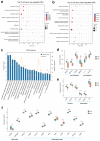
Dairy cows face metabolic challenges around the time of calving, leading to a negative energy balance and various postpartum health issues. Adipose tissue is crucial for cows during this period, as it regulates energy metabolism and supports immune function. Naringin, one of the main flavonoids in citrus fruit and their byproducts, is a potent antioxidant and anti-inflammatory phytoconstituent. The study aimed to evaluate the effects of supplemental naringin on performance, systemic inflammation, oxidative status, and adipose tissue metabolic status. A total of 36 multiparous Holstein cows (from ~21 d prepartum through 35 d postpartum) were provided a basal control (CON) diet or a CON diet containing naringin (NAR) at 30 g/d per cow. Supplemental NAR increased the yield of raw milk and milk protein, without affecting dry matter intake. Cows fed NAR showed significantly lower levels (p < 0.05) of serum non-esterified fatty acid (NEFA), C-reactive protein, IL-1β, IL-6, malonaldehyde, lipopolysaccharide (LPS), aspartate aminotransferase, and alanine aminotransferase, but increased (p < 0.05) glutathione peroxidase activity relative to those fed CON. Supplemental NAR increased (p < 0.05) adipose tissue adiponectin abundance, decreased inflammatory responses, and reduced oxidative stress. Lipidomic analysis showed that cows fed NAR had lower concentrations of ceramide species (p < 0.05) in the serum and adipose tissue than did the CON-fed cows. Adipose tissue proteomics showed that proteins related to lipolysis, ceramide biosynthesis, inflammation, and heat stress were downregulated (p < 0.05), while those related to glycerophospholipid biosynthesis and the extracellular matrix were upregulated (p < 0.05). Feeding NAR to cows may reduce the accumulation of ceramide by lowering serum levels of NEFA and LPS and increasing adiponectin expression, thereby decreasing inflammation and oxidative stress in adipose tissue, ultimately improving their systemic metabolic status. Including NAR in periparturient cows' diets improves lactational performance, reduces excessive lipolysis in adipose tissue, and decreases systemic and adipose tissue inflammation and oxidative stress. Integrating lipidomic and proteomic data revealed that reduced ceramide and increased glycerophospholipids may alleviate metabolic dysregulations in adipose tissue, which in turn benefits systemic metabolic status.
Keywords: adipose tissue; naringin; oxidative stress; systemic inflammation; transition dairy cow.
Conflict of interest statement
Author Sarula Bai was employed by the Beijing Sunlon Livestock Development Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Effects of shortening the close-up period length coupled with increased supply of metabolizable protein on performance and metabolic status of multiparous Holstein cows.J Dairy Sci. 2017 Aug;100(8):6199-6217. doi: 10.3168/jds.2016-12263. Epub 2017 May 24. J Dairy Sci. 2017. PMID: 28551194
-
Metabolic and blood acid-base responses to prepartum dietary cation-anion difference and calcium content in transition dairy cows.J Dairy Sci. 2022 Feb;105(2):1199-1210. doi: 10.3168/jds.2021-21191. Epub 2021 Dec 23. J Dairy Sci. 2022. PMID: 34955266
-
Biomarkers of inflammation, metabolism, and oxidative stress in blood, liver, and milk reveal a better immunometabolic status in peripartal cows supplemented with Smartamine M or MetaSmart.J Dairy Sci. 2014 Dec;97(12):7437-50. doi: 10.3168/jds.2013-7679. Epub 2014 Oct 3. J Dairy Sci. 2014. PMID: 25282419
-
Effect of diets enriched in n-6 or n-3 fatty acids on dry matter intake, energy balance, oxidative stress, and milk fat profile of transition cows.J Dairy Sci. 2023 Aug;106(8):5416-5432. doi: 10.3168/jds.2022-22540. Epub 2023 Jun 7. J Dairy Sci. 2023. PMID: 37296049
-
A Brief Overview of Oxidative Stress in Adipose Tissue with a Therapeutic Approach to Taking Antioxidant Supplements.Antioxidants (Basel). 2021 Apr 13;10(4):594. doi: 10.3390/antiox10040594. Antioxidants (Basel). 2021. PMID: 33924341 Free PMC article. Review.
Cited by
-
Egg exosome miR-145-5p decreases mitochondrial ROS to protect chicken embryo hepatocytes against apoptosis through targeting MAPK10.J Anim Sci Biotechnol. 2025 May 24;16(1):74. doi: 10.1186/s40104-025-01203-y. J Anim Sci Biotechnol. 2025. PMID: 40410809 Free PMC article.
References
-
- Ospina P.A., Nydam D.V., Stokol T., Overton T.R. Associations of elevated nonesterified fatty acids and β-hydroxybutyrate concentrations with early lactation reproductive performance and milk production in transition dairy cattle in the northeastern United States. J. Dairy Sci. 2010;93:1596–1603. doi: 10.3168/jds.2009-2852. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

