Calcium channel signalling at neuronal endoplasmic reticulum-plasma membrane junctions
- PMID: 38934485
- PMCID: PMC11668288
- DOI: 10.1042/BST20230819
Calcium channel signalling at neuronal endoplasmic reticulum-plasma membrane junctions
Abstract
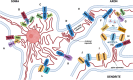
Neurons are highly specialised cells that need to relay information over long distances and integrate signals from thousands of synaptic inputs. The complexity of neuronal function is evident in the morphology of their plasma membrane (PM), by far the most intricate of all cell types. Yet, within the neuron lies an organelle whose architecture adds another level to this morphological sophistication - the endoplasmic reticulum (ER). Neuronal ER is abundant in the cell body and extends to distant axonal terminals and postsynaptic dendritic spines. It also adopts specialised structures like the spine apparatus in the postsynapse and the cisternal organelle in the axon initial segment. At membrane contact sites (MCSs) between the ER and the PM, the two membranes come in close proximity to create hubs of lipid exchange and Ca2+ signalling called ER-PM junctions. The development of electron and light microscopy techniques extended our knowledge on the physiological relevance of ER-PM MCSs. Equally important was the identification of ER and PM partners that interact in these junctions, most notably the STIM-ORAI and VAP-Kv2.1 pairs. The physiological functions of ER-PM junctions in neurons are being increasingly explored, but their molecular composition and the role in the dynamics of Ca2+ signalling are less clear. This review aims to outline the current state of research on the topic of neuronal ER-PM contacts. Specifically, we will summarise the involvement of different classes of Ca2+ channels in these junctions, discuss their role in neuronal development and neuropathology and propose directions for further research.
Keywords: ER-PM junctions; ORAI; STIM; endoplasmic reticulum; membrane contact sites; store-operated calcium entry.
© 2024 The Author(s).
Conflict of interest statement
The authors declare that there are no competing interests associated with the manuscript.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

