Loss of transient receptor potential channel 5 causes obesity and postpartum depression
- PMID: 38959890
- PMCID: PMC11961024
- DOI: 10.1016/j.cell.2024.06.001
Loss of transient receptor potential channel 5 causes obesity and postpartum depression
Abstract
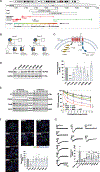
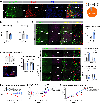
Hypothalamic neural circuits regulate instinctive behaviors such as food seeking, the fight/flight response, socialization, and maternal care. Here, we identified microdeletions on chromosome Xq23 disrupting the brain-expressed transient receptor potential (TRP) channel 5 (TRPC5). This family of channels detects sensory stimuli and converts them into electrical signals interpretable by the brain. Male TRPC5 deletion carriers exhibited food seeking, obesity, anxiety, and autism, which were recapitulated in knockin male mice harboring a human loss-of-function TRPC5 mutation. Women carrying TRPC5 deletions had severe postpartum depression. As mothers, female knockin mice exhibited anhedonia and depression-like behavior with impaired care of offspring. Deletion of Trpc5 from oxytocin neurons in the hypothalamic paraventricular nucleus caused obesity in both sexes and postpartum depressive behavior in females, while Trpc5 overexpression in oxytocin neurons in knock-in mice reversed these phenotypes. We demonstrate that TRPC5 plays a pivotal role in mediating innate human behaviors fundamental to survival, including food seeking and maternal care.
Keywords: TRP channels; anxiety; autism; hypothalamus; innate behavior; obesity; postpartum depression; sensory signaling.
Copyright © 2024 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests I.S.F. has consulted for a number of companies developing weight loss drugs (including Eli Lilly, Novo Nordisk, and Rhythm Pharmaceuticals) and investors (Goldman Sachs, SV Health). I.S.F. is a member of the Advisory Board of Cell. J.R.B.P. is an employee and shareholder of Insmed, receives research funding from GSK, and is a paid consultant for WW International. K.W.W. holds shares in Novo Nordisk and Eli Lilly.
Figures





References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

