Direct and indirect pathways for heterosynaptic interaction underlying developmental synapse elimination in the mouse cerebellum
- PMID: 38961250
- PMCID: PMC11222442
- DOI: 10.1038/s42003-024-06447-4
Direct and indirect pathways for heterosynaptic interaction underlying developmental synapse elimination in the mouse cerebellum
Abstract
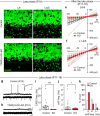
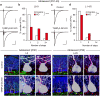
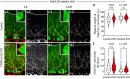
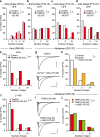
Developmental synapse elimination is crucial for shaping mature neural circuits. In the neonatal mouse cerebellum, Purkinje cells (PCs) receive excitatory synaptic inputs from multiple climbing fibers (CFs) and synapses from all but one CF are eliminated by around postnatal day 20. Heterosynaptic interaction between CFs and parallel fibers (PFs), the axons of cerebellar granule cells (GCs) forming excitatory synapses onto PCs and molecular layer interneurons (MLIs), is crucial for CF synapse elimination. However, mechanisms for this heterosynaptic interaction are largely unknown. Here we show that deletion of AMPA-type glutamate receptor functions in GCs impairs CF synapse elimination mediated by metabotropic glutamate receptor 1 (mGlu1) signaling in PCs. Furthermore, CF synapse elimination is impaired by deleting NMDA-type glutamate receptors from MLIs. We propose that PF activity is crucial for CF synapse elimination by directly activating mGlu1 in PCs and indirectly enhancing the inhibition of PCs through activating NMDA receptors in MLIs.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Reduced GABAergic inhibition and impaired synapse elimination by neuroligin-2 deletion from Purkinje cells of the developing cerebellum.Front Neural Circuits. 2025 Mar 14;19:1530141. doi: 10.3389/fncir.2025.1530141. eCollection 2025. Front Neural Circuits. 2025. PMID: 40160866 Free PMC article.
-
Roles of glutamate receptor delta 2 subunit (GluRdelta 2) and metabotropic glutamate receptor subtype 1 (mGluR1) in climbing fiber synapse elimination during postnatal cerebellar development.J Neurosci. 2001 Dec 15;21(24):9701-12. doi: 10.1523/JNEUROSCI.21-24-09701.2001. J Neurosci. 2001. PMID: 11739579 Free PMC article.
-
Postsynaptic P/Q-type Ca2+ channel in Purkinje cell mediates synaptic competition and elimination in developing cerebellum.Proc Natl Acad Sci U S A. 2011 Jun 14;108(24):9987-92. doi: 10.1073/pnas.1101488108. Epub 2011 May 31. Proc Natl Acad Sci U S A. 2011. PMID: 21628556 Free PMC article.
-
Influence of parallel fiber-Purkinje cell synapse formation on postnatal development of climbing fiber-Purkinje cell synapses in the cerebellum.Neuroscience. 2009 Sep 1;162(3):601-11. doi: 10.1016/j.neuroscience.2008.12.037. Epub 2008 Dec 31. Neuroscience. 2009. PMID: 19166909 Review.
-
Retrograde signaling for climbing fiber synapse elimination.Cerebellum. 2015 Feb;14(1):4-7. doi: 10.1007/s12311-014-0615-y. Cerebellum. 2015. PMID: 25338972 Review.
Cited by
-
Molecular and cellular mechanisms of developmental synapse elimination in the cerebellum: Involvement of autism spectrum disorder-related genes.Proc Jpn Acad Ser B Phys Biol Sci. 2024;100(9):508-523. doi: 10.2183/pjab.100.034. Proc Jpn Acad Ser B Phys Biol Sci. 2024. PMID: 39522973 Free PMC article. Review.
-
Non-allometric expansion and enhanced compartmentalization of Purkinje cell dendrites in the human cerebellum.Elife. 2025 Apr 15;14:RP105013. doi: 10.7554/eLife.105013. Elife. 2025. PMID: 40231436 Free PMC article.
-
Cholecystokinin-expressing GABA neurons elicit long-term potentiation in the cortical inhibitory synapses and attenuate sound-shock associative memory.Sci Rep. 2025 Aug 25;15(1):31220. doi: 10.1038/s41598-025-17065-3. Sci Rep. 2025. PMID: 40854983 Free PMC article.
-
Non-allometric expansion and enhanced compartmentalization of Purkinje cell dendrites in the human cerebellum.bioRxiv [Preprint]. 2025 Feb 20:2024.09.09.612113. doi: 10.1101/2024.09.09.612113. bioRxiv. 2025. Update in: Elife. 2025 Apr 15;14:RP105013. doi: 10.7554/eLife.105013. PMID: 39554002 Free PMC article. Updated. Preprint.
-
Reduced GABAergic inhibition and impaired synapse elimination by neuroligin-2 deletion from Purkinje cells of the developing cerebellum.Front Neural Circuits. 2025 Mar 14;19:1530141. doi: 10.3389/fncir.2025.1530141. eCollection 2025. Front Neural Circuits. 2025. PMID: 40160866 Free PMC article.
References
-
- Crepel F. Regression of functional synapses in the immature mammalian cerebellum. Trends Neurosci. 1982;5:266–269. doi: 10.1016/0166-2236(82)90168-0. - DOI
Publication types
MeSH terms
Substances
Grants and funding
- 18H04012/MEXT | Japan Society for the Promotion of Science (JSPS)
- 21H04785/MEXT | Japan Society for the Promotion of Science (JSPS)
- 20H05915/MEXT | Japan Society for the Promotion of Science (JSPS)
- 20K06862/MEXT | Japan Society for the Promotion of Science (JSPS)
- 20H05916/MEXT | Japan Society for the Promotion of Science (JSPS)
LinkOut - more resources
Full Text Sources

