Estrogen deprivation and estrogen receptor α antagonism decrease DSS colitis in female mice
- PMID: 38961539
- PMCID: PMC11222167
- DOI: 10.1002/prp2.1234
Estrogen deprivation and estrogen receptor α antagonism decrease DSS colitis in female mice
Abstract
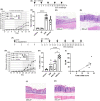
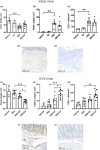
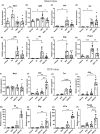
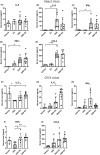
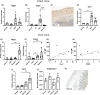
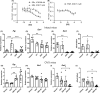
The association of hormonal contraception with increased risk of inflammatory bowel disease (IBD) observed in females suggests involvement of ovarian hormones, such as estradiol, and the estrogen receptors in the progression of intestinal inflammation. Here, we investigated the effects of prophylactic SERM2 and estradiol supplementation in dextran sulfate sodium-induced colitis using mice with intact ovaries and ovariectomized (OVX) female mice. We found that graded colitis score was threefold reduced in the OVX mice, compared to mice with intact ovaries. Estradiol supplementation, however, aggravated the colitis in OVX mice, increasing the colitis score to a similar level than what was observed in the intact mice. Further, we observed that immune infiltration and gene expression of inflammatory interleukins Il1b, Il6, and Il17a were up to 200-fold increased in estradiol supplemented OVX colitis mice, while a mild but consistent decrease was observed by SERM2 treatment in intact animals. Additionally, cyclo-oxygenase 2 induction was increased in the colon of colitis mice, in correlation with increased serum estradiol levels. Measured antagonist properties of SERM2, together with the other results presented here, indicates an exaggerating role of ERα signaling in colitis. Our results contribute to the knowledge of ovarian hormone effects in colitis and encourage further research on the potential use of ER antagonists in the colon, in order to alleviate inflammation.
Keywords: colitis; dextran sulfate; estradiol; estrogen; estrogen receptor modulators; inflammatory bowel diseases; receptors.
© 2024 The Author(s). Pharmacology Research & Perspectives published by British Pharmacological Society and American Society for Pharmacology and Experimental Therapeutics and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Effects of 17β-estradiol on colorectal cancer development after azoxymethane/dextran sulfate sodium treatment of ovariectomized mice.Biochem Pharmacol. 2019 Jun;164:139-151. doi: 10.1016/j.bcp.2019.04.011. Epub 2019 Apr 11. Biochem Pharmacol. 2019. PMID: 30981879
-
Sex steroid regulation of macrophage migration inhibitory factor in normal and inflamed colon in the female rat.Gastroenterology. 2007 Mar;132(3):982-93. doi: 10.1053/j.gastro.2006.12.028. Epub 2006 Dec 17. Gastroenterology. 2007. PMID: 17324399
-
Sarcodon aspratus Extract Ameliorates Dextran Sulfate Sodium-Induced Colitis in Mouse Colon and Mesenteric Lymph Nodes.J Food Sci. 2016 May;81(5):H1301-8. doi: 10.1111/1750-3841.13297. Epub 2016 Apr 13. J Food Sci. 2016. PMID: 27074537
-
Novel Selective Estrogen Receptor Modulator Ameliorates Murine Colitis.Int J Mol Sci. 2019 Jun 20;20(12):3007. doi: 10.3390/ijms20123007. Int J Mol Sci. 2019. PMID: 31226730 Free PMC article.
-
Effects of 17β-Estradiol on Colonic Permeability and Inflammation in an Azoxymethane/Dextran Sulfate Sodium-Induced Colitis Mouse Model.Gut Liver. 2018 Nov 15;12(6):682-693. doi: 10.5009/gnl18221. Gut Liver. 2018. PMID: 30400733 Free PMC article.
Cited by
-
Anorectal Remodeling in the Transitional Zone with Increased Expression of LGR5, SOX9, SOX2, and Keratin 13 and 5 in a Dextran Sodium Sulfate-Induced Mouse Model of Ulcerative Colitis.Int J Mol Sci. 2024 Nov 26;25(23):12706. doi: 10.3390/ijms252312706. Int J Mol Sci. 2024. PMID: 39684417 Free PMC article.
-
Investigating the Role and Underlying Mechanisms of 18β-Glycyrrhetinic Acid in the Therapy of Ulcerative Colitis Through Modulation of the PPAR-γ/NF-κB Signaling Pathway.J Inflamm Res. 2025 Jun 10;18:7529-7543. doi: 10.2147/JIR.S510949. eCollection 2025. J Inflamm Res. 2025. PMID: 40519656 Free PMC article.
References
-
- Wang R, Li Z, Liu S, Zhang D. Global, regional and national burden of inflammatory bowel disease in 204 countries and territories from 1990 to 2019: a systematic analysis based on the global burden of disease study 2019. BMJ Open. 2023;13(3):e065186. doi:10.1136/bmjopen-2022-065186 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

