Hydroxychloroquine is associated with lower seroconversion upon 17DD-Yellow fever primovaccination in patients with primary Sjögren's syndrome
- PMID: 38961639
- PMCID: PMC11225917
- DOI: 10.1080/21645515.2024.2318814
Hydroxychloroquine is associated with lower seroconversion upon 17DD-Yellow fever primovaccination in patients with primary Sjögren's syndrome
Abstract
The present study aimed at investigating whether the hydroxychloroquine (HCQ) treatment would impact the neutralizing antibody production, viremia levels and the kinetics of serum soluble mediators upon planned 17DD-Yellow Fever (YF) primovaccination (Bio-Manguinhos-FIOCRUZ) of primary Sjögren's syndrome (pSS). A total of 34 pSS patients and 23 healthy controls (HC) were enrolled. The pSS group was further categorized according to the use of HCQ (HCQ and Non-HCQ). The YF-plaque reduction neutralization test (PRNT ≥1:50), YF viremia (RNAnemia) and serum biomarkers analyses were performed at baseline and subsequent time-points (Day0/Day3-4/Day5-6/Day7/Day14-D28). The pSS group showed PRNT titers and seropositivity rates similar to those observed for HC (GeoMean = 238 vs 440, p = .11; 82% vs 96%, p = .13). However, the HCQ subgroup exhibited lower seroconversion rates as compared to HC (GeoMean = 161 vs 440, p = .04; 69% vs 96%, p = .02) and Non-HQC (GeoMean = 161 vs 337, p = .582; 69% vs 94%, p = .049). No differences in YF viremia were observed amongst subgroups. Serum biomarkers analyses demonstrated that HCQ subgroup exhibited increased levels of CCL2, CXL10, IL-6, IFN-γ, IL1-Ra, IL-9, IL-10, and IL-2 at baseline and displayed a consistent increase of several biomarkers along the kinetics timeline up to D14-28. These results indicated that HCQ subgroup exhibited a deficiency in assembling YF-specific immune response elicited by 17DD-YF primovaccination as compared to Non-HCQ subgroup. Our findings suggested that hydroxychloroquine is associated with a decrease in the humoral immune response after 17DD-YF primovaccination.
Keywords: 17DD-YF vaccine; Primary Sjögren’s syndrome; humoral immunity; hydroxychloroquine; serum biomarkers.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures


 , D28 =
, D28 =  , n = 34) and healthy controls (HC, D0 =
, n = 34) and healthy controls (HC, D0 =  ,D28 =
,D28 =  , n = 23). Data are shown as scattering of individual values of reciprocal of serum dilution over column charts representing geometric mean titers (95%CI). The seropositivity rates are shown as frequency (%) of subjects with titers higher than 1:50 (dashed line).Comparative analysis of PRNT titers and seropositive rates (pSS vs HC) were assessed by Student t test and Chi-square test, respectively. In all cases, significant differences were considered at p < .05.
, n = 23). Data are shown as scattering of individual values of reciprocal of serum dilution over column charts representing geometric mean titers (95%CI). The seropositivity rates are shown as frequency (%) of subjects with titers higher than 1:50 (dashed line).Comparative analysis of PRNT titers and seropositive rates (pSS vs HC) were assessed by Student t test and Chi-square test, respectively. In all cases, significant differences were considered at p < .05.
 , D28 =
, D28 =  , n = 18) or HCQ (D0 =
, n = 18) or HCQ (D0 =  , D28 =
, D28 =  , n = 16) and samples from healthy controls (HC, D0 =
, n = 16) and samples from healthy controls (HC, D0 =  , D28 =
, D28 =  , n = 23). Data are shown as scattering of individual values of reciprocal of serum dilution over column charts representing geometric mean titers (95% CI). The seropositivity rates are shown as frequency (%) of subjects with titers higher than 1:50 (dashed line). Comparative analysis of PRNT titers and seropositive rates (pSS subgroups vs HC) were assessed by Student t test and Chi-square test, respectively. In all cases, significant differences were considered at p < .05 and highlighted by bold format.
, n = 23). Data are shown as scattering of individual values of reciprocal of serum dilution over column charts representing geometric mean titers (95% CI). The seropositivity rates are shown as frequency (%) of subjects with titers higher than 1:50 (dashed line). Comparative analysis of PRNT titers and seropositive rates (pSS subgroups vs HC) were assessed by Student t test and Chi-square test, respectively. In all cases, significant differences were considered at p < .05 and highlighted by bold format.
 , n = 18) or HCQ (
, n = 18) or HCQ ( , n = 16) and healthy controls (HC,
, n = 16) and healthy controls (HC,  , n = 23) at distinct time points upon 17DD-YF primovaccination (D3,D4, D5, D6, D7, and D14). Data are shown as GeoMean of copies/mL (95%CI) along the kinetics timeline. Dashed line represents the limit of detection (6.25 copies/µL). Undetectable levels were computed as 0.001 copies/mL. Viremia kinetic curves are shown as GeoMean of copies/mL in overlayed line charts. Comparative analysis of viremia levels at the day of viremia peak (gray rectangles) was carried out by ANOVA followed by Tukey’s post-test and the viremia kinetic curves compared by Kolmogorov-Smirnov (KS) test. In all cases, significant differences at were considered at p < .05.
, n = 23) at distinct time points upon 17DD-YF primovaccination (D3,D4, D5, D6, D7, and D14). Data are shown as GeoMean of copies/mL (95%CI) along the kinetics timeline. Dashed line represents the limit of detection (6.25 copies/µL). Undetectable levels were computed as 0.001 copies/mL. Viremia kinetic curves are shown as GeoMean of copies/mL in overlayed line charts. Comparative analysis of viremia levels at the day of viremia peak (gray rectangles) was carried out by ANOVA followed by Tukey’s post-test and the viremia kinetic curves compared by Kolmogorov-Smirnov (KS) test. In all cases, significant differences at were considered at p < .05.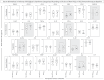
 , n = 18) or HCQ (D0 =
, n = 18) or HCQ (D0 =  , n = 16) prior 17DD-YF primovaccination. The levels of chemokines(CXCL8, CCL11, CCL3, CCL4, CCL2, CCL5, CXCL10), pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, IL-12, IFN-γ, IL-15, IL-17),regulatory cytokines (IL-1Ra, IL-4, IL-5, IL-9, IL-10, IL-13) and growth factors (FGF-basic, PDGF, VEGF, G-CSF, GM-CSF, IL-2 and IL-7) are shown as violin plot distribution (min and max) of serum concentrations (pg/mL) underscoring the median (continuous line) and 25th and 75th interquartile range (dashedlines). Significant differences at p < .05 are identified by connecting line and underscored by gray background.
, n = 16) prior 17DD-YF primovaccination. The levels of chemokines(CXCL8, CCL11, CCL3, CCL4, CCL2, CCL5, CXCL10), pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, IL-12, IFN-γ, IL-15, IL-17),regulatory cytokines (IL-1Ra, IL-4, IL-5, IL-9, IL-10, IL-13) and growth factors (FGF-basic, PDGF, VEGF, G-CSF, GM-CSF, IL-2 and IL-7) are shown as violin plot distribution (min and max) of serum concentrations (pg/mL) underscoring the median (continuous line) and 25th and 75th interquartile range (dashedlines). Significant differences at p < .05 are identified by connecting line and underscored by gray background.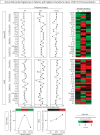
 , D5-6 =
, D5-6 =  , D14-28 =
, D14-28 =  , n = 18) or HCQ (D0 =
, n = 18) or HCQ (D0 =  , D5-6 =
, D5-6 =  , D14-28 =
, D14-28 =  , n = 16) prior 17DD-YF primovaccination. Serum biomarker signatures were assembled converting the biomarker levels originally express as continuous values (pg/mL) into categorial data, and the results reported as proportion of subjects (%) with biomarker levels above the global median cutoff defined for each serum soluble mediator. Signature curves were assembled to identify the biomarkers with proportion of subjects above the 50th percentile (dashed line). Heatmaps were constructed considering the proportion of subjects with biomarker levels above the global median cutoff at each time point (D0, D5-6 and D14-28). Color key underscores the proportion of subjects with increased biomarkers levels in percentiles: 10th (green), 50th (black) and 90th (red). Line charts show the total number of biomarkers above the global median cutoff.
, n = 16) prior 17DD-YF primovaccination. Serum biomarker signatures were assembled converting the biomarker levels originally express as continuous values (pg/mL) into categorial data, and the results reported as proportion of subjects (%) with biomarker levels above the global median cutoff defined for each serum soluble mediator. Signature curves were assembled to identify the biomarkers with proportion of subjects above the 50th percentile (dashed line). Heatmaps were constructed considering the proportion of subjects with biomarker levels above the global median cutoff at each time point (D0, D5-6 and D14-28). Color key underscores the proportion of subjects with increased biomarkers levels in percentiles: 10th (green), 50th (black) and 90th (red). Line charts show the total number of biomarkers above the global median cutoff.
 , D5-6 =
, D5-6 =  , D14-28 =
, D14-28 =  , n = 18) or HCQ (D0 =
, n = 18) or HCQ (D0 =  , D5-6 =
, D5-6 =  , D14-28 =
, D14-28 =  , n=16) prior 17DD-YF primovaccination. The integrative networks were assembled based on Pearson and Spearman “r” scores between chemokines (
, n=16) prior 17DD-YF primovaccination. The integrative networks were assembled based on Pearson and Spearman “r” scores between chemokines ( = CXCL8, CCL11, CCL3, CCL4, CCL2, CCL5, CXCL10), pro-inflammatory cytokines (
= CXCL8, CCL11, CCL3, CCL4, CCL2, CCL5, CXCL10), pro-inflammatory cytokines ( = IL-1β, IL-6, TNF-α, IL-12, IFN-γ, IL-15, IL-17), regulatory cytokines (
= IL-1β, IL-6, TNF-α, IL-12, IFN-γ, IL-15, IL-17), regulatory cytokines ( = IL-1Ra, IL-4, IL-5, IL-9, IL-10, IL-13) and growth factors (
= IL-1Ra, IL-4, IL-5, IL-9, IL-10, IL-13) and growth factors ( = FGF-basic, PDGF, VEGF, G-CSF, GM-CSF, IL-2 and IL-7). The networks were created considering only significant correlations (p < .05). Connecting edges show weak/moderate (0.1 to 0.67, thin lines) and strong “r” scores (≥ 0.67, bold lines) between pairs of attributes. Line charts show the total number of connections between the biomarkers.
= FGF-basic, PDGF, VEGF, G-CSF, GM-CSF, IL-2 and IL-7). The networks were created considering only significant correlations (p < .05). Connecting edges show weak/moderate (0.1 to 0.67, thin lines) and strong “r” scores (≥ 0.67, bold lines) between pairs of attributes. Line charts show the total number of connections between the biomarkers.Similar articles
-
Serum biomarker profile orchestrating the seroconversion status of patients with autoimmune diseases upon planned primary 17DD Yellow fever vaccination.Sci Rep. 2021 May 17;11(1):10431. doi: 10.1038/s41598-021-89770-8. Sci Rep. 2021. PMID: 34001945 Free PMC article.
-
Previous biological therapy and impairment of the IFN-γ/IL-10 axis are associated with low immune response to 17DD-YF vaccination in patients with spondyloarthritis.Vaccine. 2022 Jul 30;40(32):4580-4593. doi: 10.1016/j.vaccine.2022.05.071. Epub 2022 Jun 18. Vaccine. 2022. PMID: 35728990
-
Planned Yellow Fever Primary Vaccination Is Safe and Immunogenic in Patients With Autoimmune Diseases: A Prospective Non-interventional Study.Front Immunol. 2020 Jul 17;11:1382. doi: 10.3389/fimmu.2020.01382. eCollection 2020. Front Immunol. 2020. PMID: 32765496 Free PMC article.
-
Immunogenicity and duration of protection after yellow fever vaccine in people living with human immunodeficiency virus: a systematic review.Clin Microbiol Infect. 2021 Jul;27(7):958-967. doi: 10.1016/j.cmi.2021.03.004. Epub 2021 Apr 1. Clin Microbiol Infect. 2021. PMID: 33813107
-
Hydroxychloroquine and Sjögren's disease: current evidences for its use.Joint Bone Spine. 2025 Jan;92(1):105799. doi: 10.1016/j.jbspin.2024.105799. Epub 2024 Oct 28. Joint Bone Spine. 2025. PMID: 39490567 Review.
Cited by
-
Explore the possible influence of Sjogren's syndrome on thyroid cancer: A literature data mining and meta-analysis.PLoS One. 2025 Feb 10;20(2):e0318747. doi: 10.1371/journal.pone.0318747. eCollection 2025. PLoS One. 2025. PMID: 39928612 Free PMC article.
References
-
- Tonacio AC, Do Nascimento Pedrosa T, Borba EF, Aikawa NE, Pasoto SG, Filho JCRF, Sampaio Barros MM, Leon EP, Lombardi SCFS, Junior AM, et al. Immunogenicity and safety of primary fractional-dose yellow fever vaccine in autoimmune rheumatic diseases. PloS Negl Trop Dis. 2021;15(11):e0010002. [accessed 2021 Nov 29]. doi:10.1371/journal.pntd.0010002. - DOI - PMC - PubMed
-
- Valim V, Machado KLLL, Miyamoto ST, Pinto AD, Rocha PCM, Serrano EV, Dinis VG, Gouvêa SA, Dias JGF, Campi-Azevedo AC. et al. Planned yellow fever Primovaccination is Safe and immunogenic in patients with autoimmune diseases: a prospective non-interventional study. Front Immunol. 2020;11:1382. [accessed 2020 Jul 17]. doi:10.3389/fimmu.2020.01382. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
