Radiopaque FeMnN-Mo composite drawn filled tubing wires for braided absorbable neurovascular devices
- PMID: 38962657
- PMCID: PMC11220465
- DOI: 10.1016/j.bioactmat.2024.06.002
Radiopaque FeMnN-Mo composite drawn filled tubing wires for braided absorbable neurovascular devices
Abstract
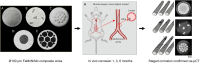
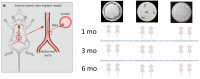
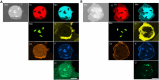
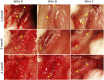
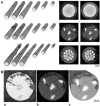
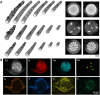
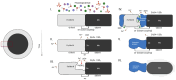
Flow diverter devices are small stents used to divert blood flow away from aneurysms in the brain, stagnating flow and inducing intra-aneurysmal thrombosis which in time will prevent aneurysm rupture. Current devices are formed from thin (∼25 μm) wires which will remain in place long after the aneurysm has been mitigated. As their continued presence could lead to secondary complications, an absorbable flow diverter which dissolves into the body after aneurysm occlusion is desirable. The absorbable metals investigated to date struggle to achieve the necessary combination of strength, elasticity, corrosion rate, fragmentation resistance, radiopacity, and biocompatibility. This work proposes and investigates a new composite wire concept combining absorbable iron alloy (FeMnN) shells with one or more pure molybdenum (Mo) cores. Various wire configurations are produced and drawn to 25-250 μm wires. Tensile testing revealed high and tunable mechanical properties on par with existing flow diverter materials. In vitro degradation testing of 100 μm wire in DMEM to 7 days indicated progressive corrosion and cracking of the FeMnN shell but not of the Mo, confirming the cathodic protection of the Mo by the FeMnN and thus mitigation of premature fragmentation risk. In vivo implantation and subsequent μCT of the same wires in mouse aortas to 6 months showed meaningful corrosion had begun in the FeMnN shell but not yet in the Mo filament cores. In total, these results indicate that these composites may offer an ideal combination of properties for absorbable flow diverters.
© 2024 The Authors.
Figures





Similar articles
-
Evaluation of FeMnN alloy bioresorbable flow diverters in the rabbit elastase induced aneurysm model.Front Bioeng Biotechnol. 2025 Feb 25;13:1522696. doi: 10.3389/fbioe.2025.1522696. eCollection 2025. Front Bioeng Biotechnol. 2025. PMID: 40070550 Free PMC article.
-
Evaluation of FeMnN alloy bioresorbable flow diverting stents in the rabbit abdominal aorta.Bioact Mater. 2025 Feb 12;48:18-28. doi: 10.1016/j.bioactmat.2025.01.039. eCollection 2025 Jun. Bioact Mater. 2025. PMID: 40303967 Free PMC article.
-
Safety and Efficacy of Low-Profile Braided Stents versus Flow Diverters in the Reconstructive Technique in the Treatment of Patients with Vertebrobasilar Dolichoectasia Aneurysms: A Cohort of 47 Patients with Long-Term Follow-Up.AJNR Am J Neuroradiol. 2024 Feb 7;45(2):176-182. doi: 10.3174/ajnr.A8091. AJNR Am J Neuroradiol. 2024. PMID: 38238095 Free PMC article.
-
Current status and perspectives of zinc-based absorbable alloys for biomedical applications.Acta Biomater. 2019 Oct 1;97:1-22. doi: 10.1016/j.actbio.2019.07.034. Epub 2019 Jul 24. Acta Biomater. 2019. PMID: 31351253 Review.
-
Bioresorbable flow diverters for the treatment of intracranial aneurysms: review of current literature and future directions.J Neurointerv Surg. 2023 Feb;15(2):178-182. doi: 10.1136/neurintsurg-2022-018941. Epub 2022 May 30. J Neurointerv Surg. 2023. PMID: 35636949 Free PMC article. Review.
Cited by
-
Evaluation of FeMnN alloy bioresorbable flow diverters in the rabbit elastase induced aneurysm model.Front Bioeng Biotechnol. 2025 Feb 25;13:1522696. doi: 10.3389/fbioe.2025.1522696. eCollection 2025. Front Bioeng Biotechnol. 2025. PMID: 40070550 Free PMC article.
-
Evaluation of FeMnN alloy bioresorbable flow diverting stents in the rabbit abdominal aorta.Bioact Mater. 2025 Feb 12;48:18-28. doi: 10.1016/j.bioactmat.2025.01.039. eCollection 2025 Jun. Bioact Mater. 2025. PMID: 40303967 Free PMC article.
References
-
- Tsao C.W., Aday A.W., Almarzooq Z.I., Alonso A., Beaton A.Z., Bittencourt M.S., Boehme A.K., Buxton A.E., Carson A.P., Commodore-Mensah Y. Heart disease and stroke statistics—2022 update: a report from the American Heart Association. Circulation. 2022;145(8):e153–e639. - PubMed
-
- Ormiston J.A., Serruys P.W. Bioabsorbable coronary stents. Circulation: Cardiovascular Interventions. 2009;2(3):255–260. - PubMed
-
- Zartner P., Cesnjevar R., Singer H., Weyand M. First successful implantation of a biodegradable metal stent into the left pulmonary artery of a preterm baby. Cathet. Cardiovasc. Interv. 2005;66(4):590–594. - PubMed
-
- Park S.-J., Kang S.-J., Virmani R., Nakano M., Ueda Y. In-stent neoatherosclerosis: a final common pathway of late stent failure. J. Am. Coll. Cardiol. 2012;59(23):2051–2057. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

