A Lipid-Sensitive Spider Peptide Toxin Exhibits Selective Anti-Leukemia Efficacy through Multimodal Mechanisms
- PMID: 38962935
- PMCID: PMC11348133
- DOI: 10.1002/advs.202404937
A Lipid-Sensitive Spider Peptide Toxin Exhibits Selective Anti-Leukemia Efficacy through Multimodal Mechanisms
Abstract
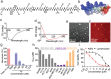
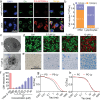
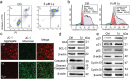
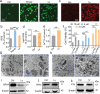
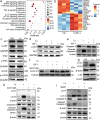
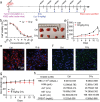
Anti-cancer peptides (ACPs) represent a promising potential for cancer treatment, although their mechanisms need to be further elucidated to improve their application in cancer therapy. Lycosin-I, a linear amphipathic peptide isolated from the venom of Lycosa singorensis, shows significant anticancer potential. Herein, it is found that Lycosin-I, which can self-assemble into a nanosphere structure, has a multimodal mechanism of action involving lipid binding for the selective and effective treatment of leukemia. Mechanistically, Lycosin-I selectively binds to the K562 cell membrane, likely due to its preferential interaction with negatively charged phosphatidylserine, and rapidly triggers membrane lysis, particularly at high concentrations. In addition, Lycosin-I induces apoptosis, cell cycle arrest in the G1 phase and ferroptosis in K562 cells by suppressing the PI3K-AKT-mTOR signaling pathway and activating cell autophagy at low concentrations. Furthermore, intraperitoneal injection of Lycosin-I inhibits tumor growth of K562 cells in a nude mouse xenograft model without causing side effects. Collectively, the multimodal effect of Lycosin-I can provide new insights into the mechanism of ACPs, and Lycosin-I, which is characterized by high potency and specificity, can be a promising lead for the development of anti-leukemia drugs.
Keywords: PI3K‐AKT‐mTOR signaling pathway; anticancer peptide; apoptosis; cell cycle arrest; ferroptosis; leukemia.
© 2024 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
MeSH terms
Substances
Grants and funding
- 2023GK3009/Subversive Innovation Competition Project of Hunan Province
- 2020RC4023/Science and Technology Innovation Program of Hunan Province
- 2023SK2096/Scientific Research Program of FuRong Laboratory
- 32201044/National Natural Science Foundation of China
- 81100360/National Natural Science Foundation of China
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
