Gut microbiota-derived metabolite trimethylamine N-oxide aggravates cognitive dysfunction induced by femoral fracture operation in mice
- PMID: 38963317
- PMCID: PMC11895615
- DOI: 10.1002/kjm2.12873
Gut microbiota-derived metabolite trimethylamine N-oxide aggravates cognitive dysfunction induced by femoral fracture operation in mice
Abstract
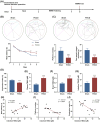
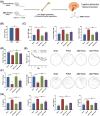
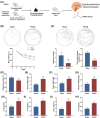
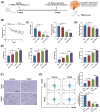
An increasing number of elderly individuals are experiencing postoperative cognitive dysfunction (POCD) problems after undergoing hip replacement surgery, with gut microbiota metabolites playing a role in its pathogenesis. Among these, the specific effects of trimethylamine N-oxide (TMAO) on POCD are still unclear. This study aimed to explore the role of TMAO on cognitive dysfunction and underlying mechanisms in mice. The POCD model was created through femoral fracture surgery in elderly mice, followed by cognitive function assessments using the Morris Water Maze and Novel Object Recognition tests. The gut microbiota depletion and fecal microbiota transplantation were performed to examine the relationship between TMAO levels and cognitive outcomes. The effects of TMAO treatment on cognitive dysfunction, microglial activation, and inflammatory cytokine levels in the brain were also evaluated, with additional assessment of the role of microglial ablation in reducing TMAO-induced cognitive impairment. Elevated TMAO levels were found to be associated with cognitive decline in mice following femoral fracture surgery, with gut microbiota depletion mitigating both TMAO elevation and cognitive dysfunction. In contrast, fecal microbiota transplantation from postoperative mice resulted in accelerated cognitive dysfunction and TMAO accumulation in germ-free mice. Furthermore, TMAO treatment worsened cognitive deficits, neuroinflammation, and promoted microglial activation, which were reversed through the ablation of microglia. TMAO exacerbates cognitive dysfunction and neuroinflammation in POCD mice, with microglial activation playing a crucial role in this process. Our findings may provide new therapeutic strategies for managing TMAO-related POCD and improving the quality of life for elderly patients.
Keywords: inflammatory cytokine; postoperative cognitive dysfunction; quality of life; trimethylamine N‐oxide; underlying mechanisms.
© 2024 The Author(s). The Kaohsiung Journal of Medical Sciences published by John Wiley & Sons Australia, Ltd on behalf of Kaohsiung Medical University.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
The presence of elevated circulating trimethylamine N-oxide exaggerates postoperative cognitive dysfunction in aged rats.Behav Brain Res. 2019 Aug 5;368:111902. doi: 10.1016/j.bbr.2019.111902. Epub 2019 Apr 10. Behav Brain Res. 2019. PMID: 30980850
-
Itaconate alleviates anesthesia/surgery-induced cognitive impairment by activating a Nrf2-dependent anti-neuroinflammation and neurogenesis via gut-brain axis.J Neuroinflammation. 2024 Apr 22;21(1):104. doi: 10.1186/s12974-024-03103-w. J Neuroinflammation. 2024. PMID: 38649932 Free PMC article.
-
Preoperative gut microbiota of POCD patients induces pre- and postoperative cognitive impairment and systemic inflammation in rats.J Neuroinflammation. 2024 Sep 12;21(1):221. doi: 10.1186/s12974-024-03220-6. J Neuroinflammation. 2024. PMID: 39267080 Free PMC article.
-
The Role of Gut Microbiota-Derived Trimethylamine N-Oxide in the Pathogenesis and Treatment of Mild Cognitive Impairment.Int J Mol Sci. 2025 Feb 6;26(3):1373. doi: 10.3390/ijms26031373. Int J Mol Sci. 2025. PMID: 39941141 Free PMC article. Review.
-
The Role of a Gut Microbial-Derived Metabolite, Trimethylamine N-Oxide (TMAO), in Neurological Disorders.Mol Neurobiol. 2022 Nov;59(11):6684-6700. doi: 10.1007/s12035-022-02990-5. Epub 2022 Aug 20. Mol Neurobiol. 2022. PMID: 35986843 Review.
Cited by
-
Associations of Microbiota and Nutrition with Cognitive Impairment in Diseases.Nutrients. 2024 Oct 21;16(20):3570. doi: 10.3390/nu16203570. Nutrients. 2024. PMID: 39458564 Free PMC article. Review.
References
-
- Bhushan S, Huang X, Duan Y, Xiao Z. The impact of regional versus general anesthesia on postoperative neurocognitive outcomes in elderly patients undergoing hip fracture surgery: a systematic review and meta‐analysis. Int J Surg. 2022;105:106854. - PubMed
-
- Xiao QX, Liu Q, Deng R, Gao ZW, Zhang Y. Postoperative cognitive dysfunction in elderly patients undergoing hip arthroplasty. Psychogeriatrics. 2020;20(4):501–509. - PubMed
-
- Tsolaki M, Sia E, Giannouli V. Anesthesia and dementia: an up‐to‐date review of the existing literature. Appl Neuropsychol Adult. 2024;31(2):181–190. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

