An atlas of brain-bone sympathetic neural circuits in mice
- PMID: 38963696
- PMCID: PMC11245306
- DOI: 10.7554/eLife.95727
An atlas of brain-bone sympathetic neural circuits in mice
Abstract
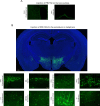
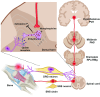
There is clear evidence that the sympathetic nervous system (SNS) mediates bone metabolism. Histological studies show abundant SNS innervation of the periosteum and bone marrow-these nerves consist of noradrenergic fibers that immunostain for tyrosine hydroxylase, dopamine beta-hydroxylase, or neuropeptide Y. Nonetheless, the brain sites that send efferent SNS outflow to the bone have not yet been characterized. Using pseudorabies (PRV) viral transneuronal tracing, we report, for the first time, the identification of central SNS outflow sites that innervate bone. We find that the central SNS outflow to bone originates from 87 brain nuclei, sub-nuclei, and regions of six brain divisions, namely the midbrain and pons, hypothalamus, hindbrain medulla, forebrain, cerebral cortex, and thalamus. We also find that certain sites, such as the raphe magnus (RMg) of the medulla and periaqueductal gray (PAG) of the midbrain, display greater degrees of PRV152 infection, suggesting that there is considerable site-specific variation in the levels of central SNS outflow to the bone. This comprehensive compendium illustrating the central coding and control of SNS efferent signals to bone should allow for a greater understanding of the neural regulation of bone metabolism, and importantly and of clinical relevance, mechanisms for central bone pain.
Keywords: bone pain; brain-bone connection; central regulation of bone metabolism; medicine; mouse; viral tracing.
© 2024, Ryu et al.
Conflict of interest statement
VR, DL, KG, SK Reviewing editor, eLife, AG, RW, FK, LC, HK, OM, OB, SS No competing interests declared, TY Senior editor, eLife, MZ consults for Gershon Lehmann, Guidepoint and Coleman groups
Figures

Update of
-
An Atlas of Brain-Bone Sympathetic Neural Circuits.bioRxiv [Preprint]. 2024 Feb 8:2024.02.07.579382. doi: 10.1101/2024.02.07.579382. bioRxiv. 2024. Update in: Elife. 2024 Jul 04;13:e95727. doi: 10.7554/eLife.95727. PMID: 38370676 Free PMC article. Updated. Preprint.
Similar articles
-
An Atlas of Brain-Bone Sympathetic Neural Circuits.bioRxiv [Preprint]. 2024 Feb 8:2024.02.07.579382. doi: 10.1101/2024.02.07.579382. bioRxiv. 2024. Update in: Elife. 2024 Jul 04;13:e95727. doi: 10.7554/eLife.95727. PMID: 38370676 Free PMC article. Updated. Preprint.
-
Periaqueductal gray matter input to cardiac-related sympathetic premotor neurons.Brain Res. 1998 May 11;792(2):179-92. doi: 10.1016/s0006-8993(98)00029-8. Brain Res. 1998. PMID: 9593884
-
Characterization of the central nervous system innervation of the rat spleen using viral transneuronal tracing.J Comp Neurol. 2001 Oct 8;439(1):1-18. doi: 10.1002/cne.1331. J Comp Neurol. 2001. PMID: 11579378
-
Brain-adipose tissue cross talk.Proc Nutr Soc. 2005 Feb;64(1):53-64. doi: 10.1079/pns2004409. Proc Nutr Soc. 2005. PMID: 15877923 Review.
-
The sympathetic nerve--an integrative interface between two supersystems: the brain and the immune system.Pharmacol Rev. 2000 Dec;52(4):595-638. Pharmacol Rev. 2000. PMID: 11121511 Review.
Cited by
-
Connection and communication between the nervous and immune systems.Nat Rev Immunol. 2025 Jul 10. doi: 10.1038/s41577-025-01199-6. Online ahead of print. Nat Rev Immunol. 2025. PMID: 40640567 Review.
-
Expression Profiles of lncRNAs and mRNAs in the Mouse Brain Infected with Pseudorabies Virus: A Bioinformatic Analysis.Viruses. 2025 Apr 17;17(4):580. doi: 10.3390/v17040580. Viruses. 2025. PMID: 40285022 Free PMC article.
References
-
- Adler ES, Hollis JH, Clarke IJ, Grattan DR, Oldfield BJ. Neurochemical characterization and sexual dimorphism of projections from the brain to abdominal and subcutaneous white adipose tissue in the rat. The Journal of Neuroscience. 2012;32:15913–15921. doi: 10.1523/JNEUROSCI.2591-12.2012. - DOI - PMC - PubMed
-
- Baptista-de-Souza D, Pelarin V, Canto-de-Souza L, Nunes-de-Souza RL, Canto-de-Souza A. Interplay between 5-HT2C and 5-HT1A receptors in the dorsal periaqueductal gray in the modulation of fear-induced antinociception in mice. Neuropharmacology. 2018;140:100–106. doi: 10.1016/j.neuropharm.2018.07.027. - DOI - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

