ERG Regulates Lymphatic Vessel Specification Genes and Its Deficiency Impairs Wound Healing-Associated Lymphangiogenesis
- PMID: 38965683
- PMCID: PMC11521767
- DOI: 10.1002/art.42944
ERG Regulates Lymphatic Vessel Specification Genes and Its Deficiency Impairs Wound Healing-Associated Lymphangiogenesis
Abstract
Objective: Rarefaction of blood and lymphatic vessels in the skin has been reported in systemic sclerosis (SSc) (scleroderma). E26 transformation-specific-related factor (ERG) and Friend leukemia virus-induced erythroleukemia 1 (FLI-1) are important regulators of angiogenesis, but their role in lymphatic vasculature is lesser known. The goal of this study was to determine the role of ERG and FLI-1 in postnatal lymphangiogenesis and SSc lymphatic system defects.
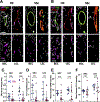
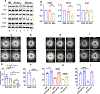
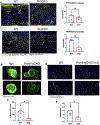
Methods: Immunofluorescence was used to detect ERG and FLI-1 in skin biopsy samples from patients with SSc and healthy controls. Transcriptional analysis of ERG or FLI-1-silenced human dermal lymphatic endothelial cells (LECs) was performed using microarrays. Effects of ERG and FLI-1 deficiency on in vitro tubulogenesis in human dermal LECs were examined using a Matrigel assay. ERG and FLI-1 endothelial-specific knockouts and ERG lymphatic-specific knockouts were generated to examine vessel regeneration in mice.
Results: ERG and FLI-1 protein levels were reduced in the blood and lymphatic vasculature in SSc skin biopsy samples. ERG levels were shown to regulate genes involved in lymphatic vessel specification, including vascular endothelial growth factor receptor 3/FLT-4, lymphatic vessel endothelial hyaluronan receptor 1, SOX-18, and prospero homeobox 1 (PROX-1), whereas FLI-1 enhanced the function of ERG. The ERG-FLT-4 pathway regulated in vitro tubulogenesis in human LECs. Deficiency of ERG or FLI-1 similarly impaired the function of blood vessels in mice. However, only ERG deficiency affected the regeneration of lymphatic vessels during wound healing.
Conclusion: ERG and FLI-1 are essential regulators of blood and lymphatic vessel regeneration. Deficiency of ERG and FLI-1 in SSc endothelial cells may contribute to the impairment of blood and lymphatic vasculature in patients with SSc.
© 2024 American College of Rheumatology.
Figures



References
-
- Wong BW, Zecchin A, Garcia-Caballero M, Carmeliet P. Emerging Concepts in Organ-Specific Lymphatic Vessels and Metabolic Regulation of Lymphatic Development. Dev Cell. 2018;45(3):289–301. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

