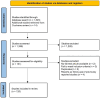
The potential of new nicotine and tobacco products as tools for people who smoke to quit combustible cigarettes - a systematic review of common practices and guidance towards a robust study protocol to measure cessation efficacy
- PMID: 38970058
- PMCID: PMC11225172
- DOI: 10.1186/s12954-024-01047-1
The potential of new nicotine and tobacco products as tools for people who smoke to quit combustible cigarettes - a systematic review of common practices and guidance towards a robust study protocol to measure cessation efficacy
Abstract
New types of nicotine and tobacco products like electronic cigarettes (ECs), heated tobacco products or nicotine pouches have been discussed as less harmful alternatives to combustible cigarettes and other toxic forms of tobacco products. Their harm reduction potential lay in the efficient transition away from smoking to those new products. Numerous studies addressing the cessation efficacy of ECs have been published with contradictory outcomes. Yet, a comprehensive Cochrane review concluded with high certainty on the cessation efficacy of ECs. This prompted us to perform a review to identify weaknesses in common study designs and to summarize best practices for the study design on the potential of new nicotine products as cessation aids. 120 articles retrieved from Medline were found to be eligible. Most of the studies in the field were interventional trials while observational studies played a minor role in the evaluation of smoking cessation. Efficacy was predominantly assessed for ECs in 77% of the reports while heated tobacco (17%) and non-combustible products (11%) were less frequently investigated up to now. Measures to determine the efficacy were questionnaire-based assessments as well as use documentation/prevalence and abstinence rates. Studies varied largely in their duration and sample size with medians of 3 months and 156.5 participants, respectively.With the help of this review, we identified several weaknesses in the common study designs. One major limitation in longitudinal trials was the lack of compliance measures suited to verify the use status over longer time periods, relying solely on self-reports. Moreover, the motivation of the participants to quit was rarely defined and a profound familiarization period was not taken into account for the majority of the studies. To what extent such weaknesses influence the outcome of the studies was beyond the scope of this review. We encourage researchers to consider the recommendations which resulted from this review in order to determine the abuse liability and cessation efficacy of the products in a more robust manner. Finally, we like to call attention to the missing data for low- and middle-income countries which would require quitting strategies most urgently to combat the tobacco smoking epidemic.
Keywords: Compliance monitoring; Electronic cigarettes; Heated tobacco products; Nicotine pouches; Smoking abstinence; Smoking cessation efficacy.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Clinical study protocol on electronic cigarettes and nicotine pouches for smoking cessation in Pakistan: a randomized controlled trial.Trials. 2024 Jan 2;25(1):9. doi: 10.1186/s13063-023-07876-y. Trials. 2024. PMID: 38167206 Free PMC article.
-
Comparing the Effectiveness, Tolerability, and Acceptability of Heated Tobacco Products and Refillable Electronic Cigarettes for Cigarette Substitution (CEASEFIRE): Randomized Controlled Trial.JMIR Public Health Surveill. 2023 Apr 4;9:e42628. doi: 10.2196/42628. JMIR Public Health Surveill. 2023. PMID: 37014673 Free PMC article. Clinical Trial.
-
Effects of reduced-risk nicotine-delivery products on smoking prevalence and cigarette sales: an observational study.Public Health Res (Southampt). 2023 Sep;11(7):1-39. doi: 10.3310/RPDN7327. Public Health Res (Southampt). 2023. PMID: 37795840
-
E-cigarettes, synthetic nicotine, heated-tobacco and smokeless nicotine delivery products: the nicotine landscape beyond combustible cigarettes.Swiss Med Wkly. 2024 Nov 4;154:3583. doi: 10.57187/s.3583. Swiss Med Wkly. 2024. PMID: 39835719 Review.
-
Different doses, durations and modes of delivery of nicotine replacement therapy for smoking cessation.Cochrane Database Syst Rev. 2019 Apr 18;4(4):CD013308. doi: 10.1002/14651858.CD013308. Cochrane Database Syst Rev. 2019. Update in: Cochrane Database Syst Rev. 2023 Jun 19;6:CD013308. doi: 10.1002/14651858.CD013308.pub2. PMID: 30997928 Free PMC article. Updated.
Cited by
-
Observational studies of exposure to tobacco and nicotine products: Best practices for maximizing statistical precision and accuracy.iScience. 2025 Feb 8;28(3):111985. doi: 10.1016/j.isci.2025.111985. eCollection 2025 Mar 21. iScience. 2025. PMID: 40104063 Free PMC article. Review.
-
Editorial: World No-Tobacco: effects of tobacco and nicotine on the brain.Front Pharmacol. 2025 May 6;16:1610178. doi: 10.3389/fphar.2025.1610178. eCollection 2025. Front Pharmacol. 2025. PMID: 40395725 Free PMC article. No abstract available.
References
-
- GBD 2019 Tobacco Collaborators Spatial, temporal, and demographic patterns in prevalence of smoking tobacco use and attributable disease burden in 204 countries and territories, 1990–2019: a systematic analysis from the global burden of Disease Study 2019. Lancet. 2021;397:2337–60. doi: 10.1016/S0140-6736(21)01169-7. - DOI - PMC - PubMed
-
- Rigotti NA, Kruse GR, Livingstone-Banks J, Hartmann-Boyce J. Treat Tob Smoking: Rev Jama. 2022;327:566–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous