Excessive accumulation of epicardial adipose tissue promotes microvascular obstruction formation after myocardial ischemia/reperfusion through modulating macrophages polarization
- PMID: 38970123
- PMCID: PMC11227217
- DOI: 10.1186/s12933-024-02342-8
Excessive accumulation of epicardial adipose tissue promotes microvascular obstruction formation after myocardial ischemia/reperfusion through modulating macrophages polarization
Abstract
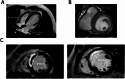
Background: Owing to its unique location and multifaceted metabolic functions, epicardial adipose tissue (EAT) is gradually emerging as a new metabolic target for coronary artery disease risk stratification. Microvascular obstruction (MVO) has been recognized as an independent risk factor for unfavorable prognosis in acute myocardial infarction patients. However, the concrete role of EAT in the pathogenesis of MVO formation in individuals with ST-segment elevation myocardial infarction (STEMI) remains unclear. The objective of the study is to evaluate the correlation between EAT accumulation and MVO formation measured by cardiac magnetic resonance (CMR) in STEMI patients and clarify the underlying mechanisms involved in this relationship.
Methods: Firstly, we utilized CMR technique to explore the association of EAT distribution and quantity with MVO formation in patients with STEMI. Then we utilized a mouse model with EAT depletion to explore how EAT affected MVO formation under the circumstances of myocardial ischemia/reperfusion (I/R) injury. We further investigated the immunomodulatory effect of EAT on macrophages through co-culture experiments. Finally, we searched for new therapeutic strategies targeting EAT to prevent MVO formation.
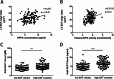
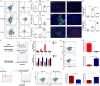
Results: The increase of left atrioventricular EAT mass index was independently associated with MVO formation. We also found that increased circulating levels of DPP4 and high DPP4 activity seemed to be associated with EAT increase. EAT accumulation acted as a pro-inflammatory mediator boosting the transition of macrophages towards inflammatory phenotype in myocardial I/R injury through secreting inflammatory EVs. Furthermore, our study declared the potential therapeutic effects of GLP-1 receptor agonist and GLP-1/GLP-2 receptor dual agonist for MVO prevention were at least partially ascribed to its impact on EAT modulation.
Conclusions: Our work for the first time demonstrated that excessive accumulation of EAT promoted MVO formation by promoting the polarization state of cardiac macrophages towards an inflammatory phenotype. Furthermore, this study identified a very promising therapeutic strategy, GLP-1/GLP-2 receptor dual agonist, targeting EAT for MVO prevention following myocardial I/R injury.
Keywords: Epicardial adipose tissue; GLP-1/GLP-2 receptor dual agonist; Inflammation; Liraglutide; Macrophage; Microvascular obstruction.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

