Comprehensive characterization of the neurogenic and neuroprotective action of a novel TrkB agonist using mouse and human stem cell models of Alzheimer's disease
- PMID: 38971770
- PMCID: PMC11227723
- DOI: 10.1186/s13287-024-03818-w
Comprehensive characterization of the neurogenic and neuroprotective action of a novel TrkB agonist using mouse and human stem cell models of Alzheimer's disease
Abstract
Background: Neural stem cell (NSC) proliferation and differentiation in the mammalian brain decreases to minimal levels postnatally. Nevertheless, neurogenic niches persist in the adult cortex and hippocampus in rodents, primates and humans, with adult NSC differentiation sharing key regulatory mechanisms with development. Adult neurogenesis impairments have been linked to Alzheimer's disease (AD) pathology. Addressing these impairments by using neurotrophic factors is a promising new avenue for therapeutic intervention based on neurogenesis. However, this possibility has been hindered by technical difficulties of using in-vivo models to conduct screens, including working with scarce NSCs in the adult brain and differences between human and mouse models or ethical limitations.
Methods: Here, we use a combination of mouse and human stem cell models for comprehensive in-vitro characterization of a novel neurogenic compound, focusing on the brain-derived neurotrophic factor (BDNF) pathway. The ability of ENT-A011, a steroidal dehydroepiandrosterone derivative, to activate the tyrosine receptor kinase B (TrkB) receptor was tested through western blotting in NIH-3T3 cells and its neurogenic and neuroprotective action were assessed through proliferation, cell death and Amyloid-β (Aβ) toxicity assays in mouse primary adult hippocampal NSCs, mouse embryonic cortical NSCs and neural progenitor cells (NPCs) differentiated from three human induced pluripotent stem cell lines from healthy and AD donors. RNA-seq profiling was used to assess if the compound acts through the same gene network as BDNF in human NPCs.
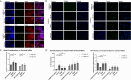
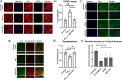
Results: ENT-A011 was able to increase proliferation of mouse primary adult hippocampal NSCs and embryonic cortical NSCs, in the absence of EGF/FGF, while reducing Aβ-induced cell death, acting selectively through TrkB activation. The compound was able to increase astrocytic gene markers involved in NSC maintenance, protect hippocampal neurons from Αβ toxicity and prevent synapse loss after Aβ treatment. ENT-A011 successfully induces proliferation and prevents cell death after Aβ toxicity in human NPCs, acting through a core gene network shared with BDNF as shown through RNA-seq.
Conclusions: Our work characterizes a novel BDNF mimetic with preferable pharmacological properties and neurogenic and neuroprotective actions in Alzheimer's disease via stem cell-based screening, demonstrating the promise of stem cell systems for short-listing competitive candidates for further testing.
Keywords: Alzheimer’s disease; BDNF; In-vitro drug testing; Neurogenesis; Stem-cell based screening; TrkB; iPSC.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

