Telescoping a Prenyltransferase and a Diterpene Synthase to Transform Unnatural FPP Derivatives to Diterpenoids
- PMID: 38976793
- PMCID: PMC11267608
- DOI: 10.1021/acs.orglett.4c01670
Telescoping a Prenyltransferase and a Diterpene Synthase to Transform Unnatural FPP Derivatives to Diterpenoids
Abstract
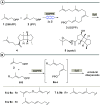
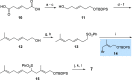
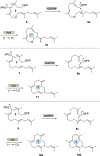
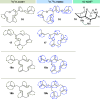
New diterpenoids are accessible from non-natural FPP derivatives as substrates for an enzymatic elongation cyclization cascade using the geranylgeranyl pyrophosphate synthase (GGPPS) from Streptomyces cyaneofuscatus and the spata-13,17-diene synthase (SpS) from Streptomyces xinghaiensis. This approach led to four new biotransformation products including three new cyclododecane cores and a macrocyclic ether. For the first time, a 1,12-terpene cyclization was observed when shifting the central olefinic double bond toward the geminial methyl groups creating a nonconjugated 1,4-diene.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Structure of bifunctional variediene synthase yields unique insight on biosynthetic diterpene assembly and cyclization.Nat Commun. 2025 Jun 2;16(1):5089. doi: 10.1038/s41467-025-60537-3. Nat Commun. 2025. PMID: 40456796 Free PMC article.
-
Methods for the preparation and analysis of the diterpene cyclase fusicoccadiene synthase.Methods Enzymol. 2024;699:89-119. doi: 10.1016/bs.mie.2023.11.003. Epub 2023 Dec 8. Methods Enzymol. 2024. PMID: 38942517 Free PMC article.
-
Chemoenzymatic Formation of Oxa-Terpenoids by Sesqui- and Diterpene Synthase-Mediated Biotransformations with 9-Oxy-FPP Ether Derivatives.Biochemistry. 2025 Jan 21;64(2):498-508. doi: 10.1021/acs.biochem.4c00589. Epub 2024 Dec 28. Biochemistry. 2025. PMID: 39731539 Free PMC article.
-
Undecaprenyl diphosphate synthase, a cis-prenyltransferase synthesizing lipid carrier for bacterial cell wall biosynthesis.Mol Membr Biol. 2012 Nov;29(7):267-73. doi: 10.3109/09687688.2012.674162. Epub 2012 Apr 4. Mol Membr Biol. 2012. PMID: 22471620 Review.
-
To gibberellins and beyond! Surveying the evolution of (di)terpenoid metabolism.Annu Rev Plant Biol. 2014;65:259-86. doi: 10.1146/annurev-arplant-050213-035705. Epub 2014 Jan 22. Annu Rev Plant Biol. 2014. PMID: 24471837 Free PMC article. Review.
Cited by
-
Synthetic Biology in Natural Product Biosynthesis.Chem Rev. 2025 Apr 9;125(7):3814-3931. doi: 10.1021/acs.chemrev.4c00567. Epub 2025 Mar 21. Chem Rev. 2025. PMID: 40116601 Review.
-
Enzyme-Catalysed Formation of Hydrocarbon Scaffolds from Geranylgeranyl Diphosphate Analogs with Shifted Double Bonds.Chemistry. 2025 Apr 22;31(23):e202500712. doi: 10.1002/chem.202500712. Epub 2025 Mar 21. Chemistry. 2025. PMID: 40067141 Free PMC article.
-
Engineering terpene synthases and their substrates for the biocatalytic production of terpene natural products and analogues.Chem Commun (Camb). 2025 Feb 4;61(12):2468-2483. doi: 10.1039/d4cc05785f. Chem Commun (Camb). 2025. PMID: 39784321 Free PMC article. Review.
References
-
- Lynen F.; Eggerer H.; Henning U.; Kessel I. Farnesyl-pyrophosphat und 3-Methyl-Δ3-butenyl-1-pyrophosphat, die biologischen Vorsstufen des Squalens. Angew. Chem. 1958, 70, 738–742. 10.1002/ange.19580702403. - DOI
-
- Ravi B. N.; Wells R. J. A series of new diterpenes from the brown alga Dipholus marginatus (Dictyotaceae). Aust. J. Chem. 1982, 35, 129–144. 10.1071/CH9820129. - DOI
-
- Gerwick W. H.; Fenical W.; van Engen D.; Clardy J. Isolation and Structure of Spatol, a Potent Inhibitor of Cell Replication from the Brown Seaweed Spatoglossum schmittii. J. Am. Chem. Soc. 1980, 102, 7991–7993. 10.1021/ja00547a055. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

