AI-based selection of individuals for supplemental MRI in population-based breast cancer screening: the randomized ScreenTrustMRI trial
- PMID: 38977914
- PMCID: PMC11405258
- DOI: 10.1038/s41591-024-03093-5
AI-based selection of individuals for supplemental MRI in population-based breast cancer screening: the randomized ScreenTrustMRI trial
Abstract
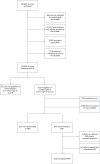
Screening mammography reduces breast cancer mortality, but studies analyzing interval cancers diagnosed after negative screens have shown that many cancers are missed. Supplemental screening using magnetic resonance imaging (MRI) can reduce the number of missed cancers. However, as qualified MRI staff are lacking, the equipment is expensive to purchase and cost-effectiveness for screening may not be convincing, the utilization of MRI is currently limited. An effective method for triaging individuals to supplemental MRI screening is therefore needed. We conducted a randomized clinical trial, ScreenTrustMRI, using a recently developed artificial intelligence (AI) tool to score each mammogram. We offered trial participation to individuals with a negative screening mammogram and a high AI score (top 6.9%). Upon agreeing to participate, individuals were assigned randomly to one of two groups: those receiving supplemental MRI and those not receiving MRI. The primary endpoint of ScreenTrustMRI is advanced breast cancer defined as either interval cancer, invasive component larger than 15 mm or lymph node positive cancer, based on a 27-month follow-up time from the initial screening. Secondary endpoints, prespecified in the study protocol to be reported before the primary outcome, include cancer detected by supplemental MRI, which is the focus of the current paper. Compared with traditional breast density measures used in a previous clinical trial, the current AI method was nearly four times more efficient in terms of cancers detected per 1,000 MRI examinations (64 versus 16.5). Most additional cancers detected were invasive and several were multifocal, suggesting that their detection was timely. Altogether, our results show that using an AI-based score to select a small proportion (6.9%) of individuals for supplemental MRI after negative mammography detects many missed cancers, making the cost per cancer detected comparable with screening mammography. ClinicalTrials.gov registration: NCT04832594 .
© 2024. The Author(s).
Conflict of interest statement
F.S. has received speaker fees from Lunit and from Pfizer. F.S. and K.S. are shareholders of ClearScanAI AB. The remaining authors declare no competing interests.
Figures

References
-
- Crosier, M. et al. Differences in Ki67 and c-erbB2 expression between screen-detected and true interval breast cancers. Clin. Cancer Res.5, 2682–2688 (1999). - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical

