Targeted inhibition of kisspeptin neurons reverses hyperandrogenemia and abnormal hyperactive LH secretion in a preclinical mouse model of polycystic ovary syndrome
- PMID: 38978296
- PMCID: PMC11373419
- DOI: 10.1093/humrep/deae153
Targeted inhibition of kisspeptin neurons reverses hyperandrogenemia and abnormal hyperactive LH secretion in a preclinical mouse model of polycystic ovary syndrome
Abstract
Study question: Do hyperactive kisspeptin neurons contribute to abnormally high LH secretion and downstream hyperandrogenemia in polycystic ovary syndrome (PCOS)-like conditions and can inhibition of kisspeptin neurons rescue such endocrine impairments?
Summary answer: Targeted inhibition of endogenous kisspeptin neuron activity in a mouse model of PCOS reduced the abnormally hyperactive LH pulse secretion and hyperandrogenemia to healthy control levels.
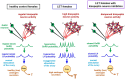
What is known already: PCOS is a reproductive disorder characterized by hyperandrogenemia, anovulation, and/or polycystic ovaries, along with a hallmark feature of abnormal LH hyper-pulsatility, but the mechanisms underlying the endocrine impairments remain unclear. A chronic letrozole (LET; aromatase inhibitor) mouse model recapitulates PCOS phenotypes, including polycystic ovaries, anovulation, high testosterone, and hyperactive LH pulses. LET PCOS-like females also have increased hypothalamic kisspeptin neuronal activation which may drive their hyperactive LH secretion and hyperandrogenemia, but this has not been tested.
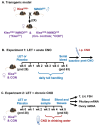
Study design, size, duration: Transgenic KissCRE+/hM4Di female mice or littermates Cre- controls were treated with placebo, or chronic LET (50 µg/day) to induce a PCOS-like phenotype, followed by acute (once) or chronic (2 weeks) clozapine-N-oxide (CNO) exposure to chemogenetically inhibit kisspeptin cells (n = 6 to 10 mice/group).
Participants/materials, setting, methods: Key endocrine measures, including in vivo LH pulse secretion patterns and circulating testosterone levels, were assessed before and after selective kisspeptin neuron inhibition and compared between PCOS groups and healthy controls. Alterations in body weights were measured and pituitary and ovarian gene expression was determined by qRT-PCR.
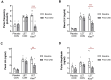
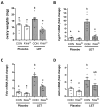
Main results and the role of chance: Acute targeted inhibition of kisspeptin neurons in PCOS mice successfully lowered the abnormally hyperactive LH pulse secretion (P < 0.05). Likewise, chronic selective suppression of kisspeptin neuron activity reversed the previously high LH and testosterone levels (P < 0.05) down to healthy control levels and rescued reproductive gene expression (P < 0. 05).
Large scale data: N/A.
Limitations, reasons for caution: Ovarian morphology was not assessed in this study. Additionally, mouse models can offer mechanistic insights into neuroendocrine processes in PCOS-like conditions but may not perfectly mirror PCOS in women.
Wider implications of the findings: These data support the hypothesis that overactive kisspeptin neurons can drive neuroendocrine PCOS-like impairments, and this may occur in PCOS women. Our findings complement recent clinical investigations using NKB receptor antagonists to lower LH in PCOS women and suggest that pharmacological dose-dependent modulation of kisspeptin neuron activity may be a valuable future therapeutic target to clinically treat hyperandrogenism and lower elevated LH in PCOS women.
Study funding/competing interest(s): This research was supported by NIH grants R01 HD111650, R01 HD090161, R01 HD100580, P50 HD012303, R01 AG078185, and NIH R24 HD102061, and a pilot project award from the British Society for Neuroendocrinology. There are no competing interests.
Keywords: GnRH; Kiss1; LH pulses; PCOS; androgen; hyperandrogenism; infertility; kisspeptin; reproduction; testosterone.
© The Author(s) 2024. Published by Oxford University Press on behalf of European Society of Human Reproduction and Embryology. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Arroyo A, Laughlin GA, Morales AJ, Yen SS. Inappropriate gonadotropin secretion in polycystic ovary syndrome: influence of adiposity. J Clin Endocrinol Metab 1997;82:3728–3733. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

