Bone-modifying agents for reducing bone loss in women with early and locally advanced breast cancer: a network meta-analysis
- PMID: 38979716
- PMCID: PMC11232105
- DOI: 10.1002/14651858.CD013451.pub2
Bone-modifying agents for reducing bone loss in women with early and locally advanced breast cancer: a network meta-analysis
Abstract
Background: Bisphosphonates and receptor activator of nuclear factor-kappa B ligand (RANKL)-inhibitors are amongst the bone-modifying agents used as supportive treatment in women with breast cancer who do not have bone metastases. These agents aim to reduce bone loss and the risk of fractures. Bisphosphonates have demonstrated survival benefits, particularly in postmenopausal women.
Objectives: To assess and compare the effects of different bone-modifying agents as supportive treatment to reduce bone mineral density loss and osteoporotic fractures in women with breast cancer without bone metastases and generate a ranking of treatment options using network meta-analyses (NMAs).
Search methods: We identified studies by electronically searching CENTRAL, MEDLINE and Embase until January 2023. We searched various trial registries and screened abstracts of conference proceedings and reference lists of identified trials.
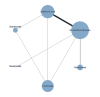
Selection criteria: We included randomised controlled trials comparing different bisphosphonates and RANKL-inihibitors with each other or against no further treatment or placebo for women with breast cancer without bone metastases.
Data collection and analysis: Two review authors independently extracted data and assessed the risk of bias of included studies and certainty of evidence using GRADE. Outcomes were bone mineral density, quality of life, overall fractures, overall survival and adverse events. We conducted NMAs and generated treatment rankings.
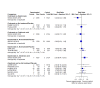
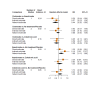
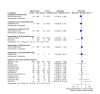
Main results: Forty-seven trials (35,163 participants) fulfilled our inclusion criteria; 34 trials (33,793 participants) could be considered in the NMA (8 different treatment options). Bone mineral density We estimated that the bone mineral density of participants with no treatment/placebo measured as total T-score was -1.34. Evidence from the NMA (9 trials; 1166 participants) suggests that treatment with ibandronate (T-score -0.77; MD 0.57, 95% CI -0.05 to 1.19) may slightly increase bone mineral density (low certainty) and treatment with zoledronic acid (T-score -0.45; MD 0.89, 95% CI 0.62 to 1.16) probably slightly increases bone mineral density compared to no treatment/placebo (moderate certainty). Risedronate (T-score -1.08; MD 0.26, 95% CI -0.32 to 0.84) may result in little to no difference compared to no treatment/placebo (low certainty). We are uncertain whether alendronate (T-score 2.36; MD 3.70, 95% CI -2.01 to 9.41) increases bone mineral density compared to no treatment/placebo (very low certainty). Quality of life No quantitative analyses could be performed for quality of life, as only three studies reported this outcome. All three studies showed only minimal differences between the respective interventions examined. Overall fracture rate We estimated that 70 of 1000 participants with no treatment/placebo had fractures. Evidence from the NMA (16 trials; 19,492 participants) indicates that treatment with clodronate or ibandronate (42 of 1000; RR 0.60, 95% CI 0.39 to 0.92; 40 of 1000; RR 0.57, 95% CI 0.38 to 0.86, respectively) decreases the number of fractures compared to no treatment/placebo (high certainty). Denosumab or zoledronic acid (51 of 1000; RR 0.73, 95% CI 0.52 to 1.01; 55 of 1000; RR 0.79, 95% CI 0.56 to 1.11, respectively) probably slightly decreases the number of fractures; and risedronate (39 of 1000; RR 0.56, 95% CI 0.15 to 2.16) probably decreases the number of fractures compared to no treatment/placebo (moderate certainty). Pamidronate (106 of 1000; RR 1.52, 95% CI 0.75 to 3.06) probably increases the number of fractures compared to no treatment/placebo (moderate certainty). Overall survival We estimated that 920 of 1000 participants with no treatment/placebo survived overall. Evidence from the NMA (17 trials; 30,991 participants) suggests that clodronate (924 of 1000; HR 0.95, 95% CI 0.77 to 1.17), denosumab (927 of 1000; HR 0.91, 95% CI 0.69 to 1.21), ibandronate (915 of 1000; HR 1.06, 95% CI 0.83 to 1.34) and zoledronic acid (925 of 1000; HR 0.93, 95% CI 0.76 to 1.14) may result in little to no difference regarding overall survival compared to no treatment/placebo (low certainty). Additionally, we are uncertain whether pamidronate (905 of 1000; HR 1.20, 95% CI 0.81 to 1.78) decreases overall survival compared to no treatment/placebo (very low certainty). Osteonecrosis of the jaw We estimated that 1 of 1000 participants with no treatment/placebo developed osteonecrosis of the jaw. Evidence from the NMA (12 trials; 23,527 participants) suggests that denosumab (25 of 1000; RR 24.70, 95% CI 9.56 to 63.83), ibandronate (6 of 1000; RR 5.77, 95% CI 2.04 to 16.35) and zoledronic acid (9 of 1000; RR 9.41, 95% CI 3.54 to 24.99) probably increases the occurrence of osteonecrosis of the jaw compared to no treatment/placebo (moderate certainty). Additionally, clodronate (3 of 1000; RR 2.65, 95% CI 0.83 to 8.50) may increase the occurrence of osteonecrosis of the jaw compared to no treatment/placebo (low certainty). Renal impairment We estimated that 14 of 1000 participants with no treatment/placebo developed renal impairment. Evidence from the NMA (12 trials; 22,469 participants) suggests that ibandronate (28 of 1000; RR 1.98, 95% CI 1.01 to 3.88) probably increases the occurrence of renal impairment compared to no treatment/placebo (moderate certainty). Zoledronic acid (21 of 1000; RR 1.49, 95% CI 0.87 to 2.58) probably increases the occurrence of renal impairment while clodronate (12 of 1000; RR 0.88, 95% CI 0.55 to 1.39) and denosumab (11 of 1000; RR 0.80, 95% CI 0.54 to 1.19) probably results in little to no difference regarding the occurrence of renal impairment compared to no treatment/placebo (moderate certainty).
Authors' conclusions: When considering bone-modifying agents for managing bone loss in women with early or locally advanced breast cancer, one has to balance between efficacy and safety. Our findings suggest that bisphosphonates (excluding alendronate and pamidronate) or denosumab compared to no treatment or placebo likely results in increased bone mineral density and reduced fracture rates. Our survival analysis that included pre and postmenopausal women showed little to no difference regarding overall survival. These treatments may lead to more adverse events. Therefore, forming an overall judgement of the best ranked bone-modifying agent is challenging. More head-to-head comparisons, especially comparing denosumab with any bisphosphonate, are needed to address gaps and validate the findings of this review.
Trial registration: ClinicalTrials.gov NCT00295646 NCT00556374 NCT00242203 NCT00525759 NCT00809484 NCT00072020 NCT02616744 NCT01077154 NCT00089661 NCT00375752 NCT00196872 NCT02682693 NCT00049452 NCT00412022 NCT00054418 NCT00512993 NCT01367288 NCT01099436 NCT00332709 NCT00009945 NCT00333229 NCT00375505 NCT00118508 NCT00485953 NCT00082277 NCT00376740 NCT00172068 NCT00127205 NCT00213980 NCT00122356 NCT00022087 NCT00107263 NCT00202059 NCT00247650 NCT00295867 NCT00324714 NCT02051218 NCT03358017 NCT03532087 NCT00050011 NCT00171340 NCT02595138 NCT00859703 NCT03691311 NCT03324932.
Copyright © 2024 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
Anne Adams: awarded a grant from the Federal Ministry of Education and Research to perform this systematic review; this did not lead to a conflict of interest, as she is a statistical editor with Cochrane Haematology, but was not involved in the editorial process for this review.
Tina Jakob: awarded a grant from the Federal Ministry of Education and Research to perform this systematic review; this did not lead to a conflict of interest.
Ina Monsef: none known; she is the Information Specialist of Cochrane Haematology, but was not involved in the editorial process for this review.
Alessandra Huth: none known.
Marco Kopp: none known.
Julia Caro‐Valenzuela: none known; a member of Cochrane Haematology, but was not involved in the editorial process for this review.
Moritz Ernst: none known; he is a member of Cochrane Haematology, but was not involved in the editorial process for this review.
Achim Wöckel: received consultancy fees to provide advice on metastatic breast cancer topics for Amgen (until end of 2020), Eli Lilly (ongoing), Hoffman‐La Roche (ongoing), Novartis (ongoing) and Pfizer (ongoing). No relevant relationships were declared in relation to early breast cancer topics.
Nicole Skoetz: none known; she is Co‐ordinating Editor of Cochrane Haematology, but was not involved in the editorial process for this review.
Figures


























Update of
References
References to studies included in this review
ABCSG‐12 2011 {published data only}
-
- Beltran-Bless AA, Clemons MJ, Fesl C, Greil R, Pond GR, Balic M, et al. Does the number of 6-monthly adjuvant zoledronate infusions received affect treatment efficacy for early breast cancer? A sub-study of ABCSG-12. European Journal of Cancer 2022;180:108-16. - PubMed
-
- Fox KR. Adding zoledronic acid to endocrine therapy in the adjuvant treatment of hormone-sensitive breast cancer in premenopausal women: a new care standard or a provocative idea? Current Oncology Reports 2010;12:1-3. - PubMed
-
- Gnant M, Dubsky P, Singer C, Greil R, Mlineritsch B, Stoeger H, et al. Bone-targeted therapy with zoledronic acid combined with adjuvant ovarian suppression plus tamoxifen or anastrozole: 62-month outcomes from the ABCSG-12 trial in premenopausal women with endocrine-responsive early breast cancer. Journal of Bone and Mineral Research 2010;25:S70.
-
- Gnant M, Jakesz R, Mlineritsch B, Luschin-Ebengreuth G, Schmid M, Menzel C, et al. Zoledronic acid effectively counteracts cancer treatment induced bone loss (CTIBL) in premenopausal breast cancer patients receiving adjuvant endocrine treatment with goserelin plus anastrozole versus goserelin plus tamoxifen - bone density subprotocol results of a randomized multicenter trial (ABCSG-12). Breast Cancer Research and Treatment 2004;Supplement:S8-9.
-
- Gnant M, Mlineritsch B, Luschin-Ebengreuth G, Kainberger F, Kaessmann H, Piswanger-Soelkner C, et al. Bone mineral density (BMD) at 5 years after diagnosis in premenopausal patients with endocrine-responsive breast cancer, after 3 years of adjuvant endocrine treatment with goserelin and tamoxifen or anastrozole or both treatments in combination with zoledronic acid - new results from ABCSG-12. Breast Cancer Research and Treatment 2007;106:S8.
ABCSG‐18 2019 {published data only}
-
- Gnant M, Frantal S, Pfeiler G, Steger GG, Egle D, Greil R, et al. Long-term outcomes of adjuvant denosumab in breast cancer: fracture reduction and survival results from 3,425 patients in the randomised, double-blind, placebocontrolled ABCSG-18 trial. Journal of Clinical Oncology 2022;40(16 Suppl):507.
-
- Gnant M, Pfeiler G, Dubsky PC, Hubalek M, Greil R, Jakesz R, et al. Adjuvant denosumab in breast cancer (ABCSG-18): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 2015;386(9992):433-43. - PubMed
-
- Gnant M, Pfeiler G, Dubsky PC, Hubalek M, Greil R, Jakesz R, et al. Adjuvant denosumab in breast cancer: results from 3,425 post-menopausal patients of the ABCSG18 trial. Journal of Clinical Oncology 2015;33(15 Suppl):504.
-
- Gnant M, Pfeiler G, Dubsky PC, Hubalek M, Greil R, Jakesz R, et al. The impact of adjuvant denosumab on disease-free survival: results from 3,425 postmenopausal patients of the ABCSG-18 trial. Cancer Research 2016;76:S2-02.
-
- Gnant M, Pfeiler G, Steger GG, Egle D, Greil R, Fitzal F, et al. Adjuvant denosumab in early breast cancer: disease-free survival analysis of 3,425 postmenopausal patients in the ABCSG-18 trial. Journal of Clinical Oncology 2018;36(15 Suppl):500.
Aft 2012 {published data only}
-
- Aft R, Chavez-MacGregor M, Trinkaus K, Naughton M, Weilbaeche RK. Effect of zoledronic acid on bone loss in women undergoing chemotherapy for breast cancer. In: 30th Annual San Antonio Breast Cancer Symposium. 2007:S38.
-
- Aft R, Naughton M, Trinkaus K, Weilbaecher K. Effect of zoledronic acid on disease-free survival and overall survival in women with clinic stage II/III undergoing neoadjuvant chemotherapy for breast cancer. Cancer Research 2010;70(24 Suppl):6-14.
-
- Aft R, Naughton M, Trinkuas K, Watson M, Weilbaecher K. Reversal of adverse effects of neoadjuvant chemotherapy on bone turnover in pre- and post-menopausal women with zoledronic acid. Journal of Clinical Oncology 2006;24(18 Suppl):16s.
ANZAC 2009 {published data only}
-
- EUCTR2007-001526-27-GB. ANZAC: A randomised phase II feasibility study investigating the biological effects of the addition of zoledronic acid to neoadjuvant comnination chemotherapy on invasive breast cancer. https://www.clinicaltrialsregister.eu/ctr-search/trial/2007-001526-27/re... (date received 2007).
-
- NCT00525759. Investigating the biological effects of the addition of zoledronic acid to pre-operative chemotherapy in breast cancer. https://clinicaltrials.gov/ct2/show/NCT00525759 (date received 6 September 2007).
-
- Wilson C, Winter MC, Holen I, Freeman JV, Evans AC, Coleman RE. The interaction between menopausal status and zoledronic acid can differentially affect serum levels of the TGFbeta superfamily. Cancer Research (35th Annual CTRC AACR San Antonio Breast Cancer Symposium. San Antonio, TX United States) 2012;72(24 Suppl):3.
-
- Winter MC, Cross SS, Ingram CE, Jolley IJ, Holen I, Hatton MQ, et al. ANZAC: A neoadjuvant biomarker study exploring the anti-tumour activity of the addition of zoledronic acid to chemotherapy in breast cancer. Annals of Oncology 2009;2:ii55.
-
- Winter MC, Evans A, Holen I, Coleman RE. The addition of zoledronic acid to combination chemotherapy decreases circulating serum levels of vascular endothelial growth factor (VEGF) in early breast cancer. Bone 2011;48(1):S30-1.
ARBI 2009 {published data only}
-
- Markopoulos C, Tzoracoleftherakis E, Koukouras D, Venizelos B, Zobolas V, Misitzis J, et al. Age effect on bone mineral density changes in breast cancer patients receiving anastrozole: results from the ARBI prospective clinical trial. Journal of Cancer Research and Clinical Oncology 2012;138(9):1569-77. - PMC - PubMed
-
- Markopoulos C, Tzorakoleftherakis E, Polychronis A, Venizelos V, Xepapadakis G, Kalogerakos K, et al. Management of bone loss in breast cancer patients: 24-month results from the ARBI trial of anastrozole with risedronate. Journal of Clinical Oncology 2009;27(15S):552.
-
- NCT00809484. Arimidex bone mass index and oral bisphosphonates (ARBI). https://clinicaltrials.gov/show/NCT00809484 (date received 17 December 2008). [CLINICALTRIALS.GOV: NCT00809484]
ARIBON 2012 {published data only}
-
- Lester J, Dodwell D, Purohit OP, Gutcher SA, Ellis SP, Thorpe R, et al. Use of monthly oral ibandronate to prevent anastrozole-induced bone loss during adjuvant treatment for breast cancer: two-year results from the ARIBON study. Journal of Clinical Oncology (meeting abstracts) 2008;26(15 Suppl):554.
-
- Lester JE, Dodwell D, Purohit OP, Gutcher SA, Ellis SP, Thorpe R, et al. Prevention of anastrozole-induced bone loss with monthly oral ibandronate during adjuvant aromatase inhibitor therapy for breast cancer. Clinical Cancer Research 2008;14(19):6336-42. - PubMed
AZURE 2018 {published data only}ISRCTN79831382
-
- ACTRN12606000293561. AZURE BR 2-03 - Adjuvant zoledronic acid in patients with high risk localised breast cancer. https://anzctr.org.au/Trial/Registration/TrialReview.aspx?ACTRN=ACTRN126... (first posted 11 July 2006).
-
- Bell R, Marshall H, Collinson M, Cameron D, Dodwell D, Keane M, et al. Reduction in fractures following adjuvant zoledronic acid in stage II/III breast cancer - the Azure trial (Big 01/04). European Journal of Cancer 2011;47:S376.
-
- Coleman R, Cameron D, Dodwell D, Bell R, Wilson C, Rathbone E, et al. Adjuvant zoledronic acid in patients with early breast cancer: final efficacy analysis of the AZURE (BIG 01/04) randomised open-label phase 3 trial. Lancet Oncology 2014;15(9):997-1006. - PubMed
-
- Coleman R, Hall A, Albanell J, Hanby A, Bell R, Cameron D, et al. Effect of MAF amplification on treatment outcomes with adjuvant zoledronic acid in early breast cancer: a secondary analysis of the international, open-label, randomised, controlled, phase 3 AZURE (BIG 01/04) trial. Lancet Oncology 2017;18(11):1543-52. - PubMed
BONADIUV 2019 {published data only}
-
- Cecchini S, Scotti V, Meattini I, De Feo ML, De Luca C, Livi L, et al. Preliminary results of "Bonadiuv" trial: a single-blind, randomized, placebo-controlled study designed to evaluate the impact of bisphosphonate treatment on bone mineral density in osteopenic women taking aromatase inhibitors. Annals of Oncology 2013;3:iii16.
-
- Livi L, Meattini I, Scotti V, Saieva C, Desideri I, Carta GA, et al. BONADIUV trial: a single blind, randomized placebo controlled phase II study using oral ibandronate for osteopenic women receiving adjuvant aromatase inhibitors: final safety analysis. Journal of Clinical Oncology 2016;34:e12043.
-
- Livi L, Saieva C, Desideri I, Scotti V, De Luca Cardillo C, Carta G, et al. A single-blind, randomized, placebo-controlled phase II study to evaluate the impact of oral ibandronate on bone mineral density in osteopenic breast cancer patients receiving adjuvant aromatase inhibitors: final results of the single-center BONADIUV trial. Cancer Research 2017;77(4 Suppl 1):P2-09-12. - PubMed
-
- Livi L, Scotti V, Desideri I, Saieva C, Cecchini S, Francolini G, et al. Phase 2 placebo-controlled, single-blind trial to evaluate the impact of oral ibandronate on bone mineral density in osteopenic breast cancer patients receiving adjuvant aromatase inhibitors: 5-year results of the single-centre BONADIUV trial. European Journal of Cancer 2019;108:100-10. - PubMed
-
- Meattini I, Scotti V, Desideri I, Saieva C, Visani L, Salvestrini V, et al. Oral ibandronate for osteopenic breast cancer patients receiving adjuvant aromatase inhibitors: secondary 5-year survival outcomes analysis of the single-center phase 2 BONADIUV trial. Cancer Research 2018;79:4.
Bundred 2009 {published data only}
-
- Bundred N, Cramer A, Morris J, Landberg G, Renshaw L, Winter M, et al. 110 Randomised placebo controlled trial studying short term biological effects of the combination of letrozole and zoledronic acid on invasive breast cancer. European Journal of Cancer 2010;8(3):90-1.
-
- Bundred N, Cramer A, Morris J, Landberg G, Renshaw L, Winter M, et al. Randomised placebo controlled trial studying short term biological effects of the combination of letrozole and zoledronic acid on invasive breast cancer. Cancer Research 2009;69(24 Suppl):2009.
-
- Bundred NJ, Landberg G, Coleman RE, Morris J, Winter MC, Holen I, et al. Short-term biological effects of zoledronic acid combined with letrozole in postmenopausal women with estrogen receptor-positive invasive breast cancer. Journal of Clinical Oncology 2009;1:e11625.
-
- EUCTR2004-004223-36-GB. Short term biological study effects of zoledronate and letrozole on invasive breast cancer - short term effects (preoperative) of femara and zometa or femara alone. https://www.clinicaltrialsregister.eu/ctr-search/search?query=eudract_nu... (first posted 1 July 2005).
Cohen 2008 {published data only}
D‐CARE 2013 {published data only}
-
- Bell R, Goss PE, Barrios CH, Finkelstein D, Iwata H, Martin M, et al. A randomised, double-blind, placebo-controlled multicentre phase 3 study comparing denosumab with placebo as adjuvant treatment for women with early-stage breast cancer who are at high risk of disease recurrence (D-care). Asia-Pacific Journal of Clinical Oncology 2011;7:150.
-
- CTRI/2010/091/001297. A phase 3 clinical trial to study the effect of drug product denosumab as adjuvant therapy for women with early-stage breast cancer, who are at high risk of disease recurrence (D-CARE ). https://ctri.nic.in/Clinicaltrials/pdf_generate.php?trialid=2089&Enc... (first posted 7 September 2010).
-
- Coleman R, Finkelstein DM, Barrios C, Martin M, Iwata H, Hegg R, et al. Adjuvant denosumab in early breast cancer (D-CARE): an international, multicentre, randomised, controlled, phase 3 trial. Lancet Oncology 2020;21(1):60-72. - PubMed
-
- Coleman RE, Barrios C, Bell R, Finkelstein DM, Iwata H, Martin M, et al. Denosumab versus placebo as adjuvant treatment for women with early-stage breast cancer at high risk of disease recurrence (D-CARE): an in progress, phase 3 clinical trial. Annals of Oncology 2012;9:ix115.
Delmas 1997 {published data only}
-
- Delmas PD, Balena R, Confravreux E, Hardouin C, Hardy P, Bremond A. Bisphosphonate risedronate prevents bone loss in women with artificial menopause due to chemotherapy of breast cancer: a double-blind, placebo-controlled study. Journal of Clinical Oncology 1997;15(3):955-62. - PubMed
Diel 1998 {published data only}
-
- Diel IJ, Solomayer EF, Costa SD, Gollan C, Goerner R, Wallwiener D, et al. Reduction in new metastases in breast cancer with adjuvant clodronate treatment. New England Journal of Medicine 1998;339(6):357-63. - PubMed
-
- Jaschke A, Bastert G, Solomayer EF, Costa S, Schuetz F, Diel IJ. Adjuvant clodronate treatment improves the overall survival of primary breast cancer patients with micrometastases to bone marrow - a longtime follow-up. Journal of Clinical Oncology 2004;22(14 Suppl):529.
Ellis 2008 {published data only}
-
- Ellis GK, Bone HG, Chlebowski R, Paul D, Spadafora S, Fan M, et al. A 24-month subgroup analysis of the effect of denosumab on bone mineral density in women with breast cancer undergoing aromatase inhibitor therapy. Cancer Research 2009;69(2 Suppl):2106. - PubMed
-
- Ellis GK, Bone HG, Chlebowski R, Paul D, Spadafora S, Fan M, et al. Effect of denosumab on bone mineral density in women receiving adjuvant aromatase inhibitors for non-metastatic breast cancer: subgroup analyses of a phase 3 study. Breast Cancer Research & Treatment 2009;118(1):81-7. - PubMed
-
- Ellis GK, Bone HG, Chlebowski R, Paul D, Spadafora S, Smith J, et al. Randomized trial of denosumab in patients receiving adjuvant aromatase inhibitors for nonmetastatic breast cancer. Journal of Clinical Oncology 2008;26(30):4875-82. - PubMed
-
- Ellis GK, Bone HG, Chlebowski RT, Paul D, Spadafora S, Smith J, et al. Subgroup analysis of a randomized, phase III study of the effect of denosumab in women with nonmetastatic breast cancer receiving aromatase inhibitor (AI) therapy [abstract no. 546]. Journal of Clinical Oncology 2008;26:17. - PubMed
-
- NCT00089661. AMG 162 in the treatment of bone loss in subjects undergoing aromatase inhibitor therapy for non-metastatic breast cancer. https://clinicaltrials.gov/ct2/show/NCT00089661 (date received 11 August 2004).
EXPAND 2011 {published data only}
-
- EUCTR2004-003888-71-DE. An open phase III trial with Letrozole (Femara®) alone or in combination with zoledronic acid (Zometa®) as extended adjuvant treatment of postmenopausal patients with primary breast cancer [EXpANd]. https://www.clinicaltrialsregister.eu/ctr-search/trial/2004-003888-71/DE (date received 16 May 2016).
-
- Hellriegel M, Mueller M, Reimer T, Baerens DT, Von Der Assen A, Hackmann J, et al. The Expand study - effect of zoledronic acid on prevention of bone loss, during extended adjuvant therapy with letrozole in postmenopausal women with primary hormone receptor positive breast cancer compared to letrozole alone. European Journal of Cancer 2011;1:S390.
FEMZONE 2014 {published data only}
-
- EUCTR2004-004007-37-DE. Neoadjuvant therapy for postmenopausal women with ER and/or PgR positive breast cancer. A randomized open phase II trial evaluating the efficacy of a 6 months preoperative treatment with Letrozole (2.5 mg/day) with or without Zoledronic acid (4 mg every 4 weeks) - FEMZONE. http://www.who.int/trialsearch/Trial2.aspx?TrialID=EUCTR2004-004007-37-DE (date received 2 March 2006).
-
- Fasching PA, Jud SM, Hauschild M, Kummel S, Schutte M, Warm M, et al. Anticancer activity of letrozole plus zoledronic acid as neoadjuvant therapy for postmenopausal patients with breast cancer: FEMZONE trial results. Cancer Research 2012;72(24 Suppl):PD07-02.
-
- Fasching PA, Jud SM, Hauschild M, Kummel S, Schutte M, Warm M, et al. Anticancer activity of letrozole plus zoledronic acid as neoadjuvant therapy for postmenopausal patients with breast cancer: FEMZONE trial results. Cancer Research 2012;72(24 Suppl):PD07-02.
-
- Lux P, Fasching P, Schulz-Wendtland R, Muth M, Beckmann MW. FEMZONE - a randomized phase II trial evaluating the efficacy of a 6 months preoperative treatment with Letrozole with or without Zoledronic acid in patients with hormone receptor positive breast cancer. Onkologie 2010;33(6):56.
GAIN 2013 {published data only}
-
- Loibl S, Conrad B, Schneeweiss A, Kreienberg R, Solomayer EF, Clemens MR, et al. Pegfilgrastim on day 2 vs. day 4 within the prospective, multi-centered GAIN study: a phase III trial to compare ETC vs. EC-TX and ibandronate vs. observation in patients with node-positive primary breast cancer (GBG 33). Support Care Cancer 2009;7(2-3):256.
-
- Mobus V, Conrad B, Schneeweis A, Kreienberg R, Solomayer EF, Clemens MR, et al. Gain study: a phase III trial to compare ETC versus EC-TX and ibandronate versus observation in patients with node-positive primary breast cancer. Journal of Clinical Oncology 2009;27(15S):568.
-
- Mobus V, Diel IJ, Harbeck N, Elling D, Jackisch C, Thomssen C, et al. GAIN Study: a phase III trial To compare ETC vs. EC-TX and ibandronate vs. observation in patients with node-positive primary breast cancer - 1st interim efficacy analysis. Cancer Research 2011;71(24 Suppl):S2-4.
-
- Mobus V, Diel IJ, Harbeck N, Elling D, Jackisch C, Thomssen C, et al. GAIN study: a phase III trial to compare ETC vs. EC-TX and ibandronate vs. observation in patients with node-positive primary breast cancer - 1st interim efficacy analysis. Cancer Research 2011;71(24 Suppl):S2-4.
-
- Mobus V, Von Minckwitz G, Jackisch C, Luck HJ, Schneeweiss A, Tesch H, et al. German Adjuvant Intergroup Node-positive study (GAIN): a phase III trial comparing two dose-dense regimens (iddEPC versus ddEC-PwX) in high-risk early breast cancer patients. Journal of Clinical Oncology 2017;28(8):1803-10. - PubMed
GeparX 2016 {published data only}
-
- Blohmer JU, Link T, Kummel S, Untch M, Just M, Fasching PA, et al. Investigating denosumab as an add-on treatment to neoadjuvant chemotherapy and two different nab-paclitaxel schedules in a 2x2 design in primary breast cancer - first results of the GeparX study. Cancer Research 2020;80(4 Suppl 1):GS3-01.
-
- EUCTR2015-001755-72-DE. Addition of denosumab to two different neoadjuvant treatment schedules of nab-paclitaxel. https://www.clinicaltrialsregister.eu/ctr-search/trial/2015-001755-72/DE (date received 2016).
-
- Kummel S, Von MG, Nekljudova V, Dan CS, Denkert C, Hanusch C, et al. Investigating denosumab as add-on neoadjuvant treatment for hormone receptor-negative, RANK-positive or RANK-negative primary breast cancer and two different nab-paclitaxel schedules - 2x2 factorial design (GeparX). Journal of Clinical Oncology 2016;34(15 Suppl):TPS635.
-
- Link T, Blohmer JU, Just M, Untch M, Stotzer O, Fasching PA. GeparX: Denosumab (Dmab) as add-on to different regimen of nab-paclitaxel (nP)-anthracycline based neoadjuvant chemotherapy (NACT) in early breast cancer (BC): subgroup analyses by RANK expression and HR status. Annals of Oncology 2020;31(Suppl 4):308-9.
Hershman 2008 {published data only}
-
- Hershman DL, McMahon D, Crew KD, Shao T, Cremers S, Brafman L, et al. Evaluation of the protective effects of zoledronic acid on bone mass in premenopausal women undergoing adjuvant chemotherapy following treatment discontinuation. Journal of Clinical Oncology 2009;27:562.
-
- Hershman DL, McMahon DJ, Crew KD, Shao T, Cremers S, Brafman L, et al. Prevention of bone loss by zoledronic acid in premenopausal women undergoing adjuvant chemotherapy persist up to one year following discontinuing treatment. Journal of Clinical Endocrinology and Metabolism 2010;95(2):559-66. - PMC - PubMed
-
- NCT00049452. Zoledronate in preventing bone loss in premenopausal women receiving chemotherapy after surgery for early stage breast cancer. https://clinicaltrials.gov/ct2/show/NCT00049452 (date received 27 January 2003).
H‐FEAT 2011 {published data only}
-
- JPRN-UMIN000005568. A randomized phase II study of efficacy of risendronate for the prevention of letrozole-induced bone mineral loss in post-menopausal breast cancer patients. http://www.who.int/trialsearch/Trial2.aspx?TrialID=JPRN-UMIN000005568 (date received 2011).
-
- Kadoya T, Masumoto N, Shigematsu H, Emi A, Kajitani K, Kobayashi Y, et al. Prevention of letrozole-induced bone loss using risedronate in postmenopausal women with hormone receptor positive breast cancer: a multicenter randomized clinical trial. Cancer Research 2016;76(4 Suppl):P1-15-03.
HOBOE 2019 {published data only}
-
- Gravina A, De Laurentis M, De Placido S, Orditura M, Orlando L, Riccardi F, et al. The HOBOE multicenter randomized phase III trial in premenopausal patients (pts) with hormonereceptor positive early breast cancer (EBC) comparing triptorelin plus either tamoxifen (T) or letrozole (L) or zoledronic acid + letrozole (ZL): 8yr efficacy analysis. Tumori 2022;108(4):2-3.
-
- Morabito A, Rossi E, Di Rella F, Esposito G, Gravina A, Labonia V, et al. Endocrine effects of adjuvant letrozole versus tamoxifen in hormone responsive postmenopausal early breast cancer patients: results from the HOBOE randomized trial. Cancer Research 2009;69(2 Suppl):1150. - PubMed
-
- NCT00412022. HOBOE: A phase 3 study of adjuvant triptorelin and tamoxifen, letrozole, or letrozole and zoledronic acid in premenopausal patients with breast cancer. https://clinicaltrials.gov/ct2/show/NCT00412022 (date received 15 December 2006).
-
- Nuzzo F, Gallo C, Lastoria S, Di Maio M, Piccirillo MC, Gravina A, et al. Bone effect of adjuvant tamoxifen, letrozole or letrozole plus zoledronic acid in early-stage breast cancer: the randomized phase 3 HOBOE study. Annals of Oncology 2012;23(8):2027-33. - PubMed
-
- Perrone F, De Laurentiis M, De Placido S, Orditura M, Cinieri S, Riccardi F, et al. Adjuvant zoledronic acid and letrozole plus ovarian function suppression in premenopausal breast cancer: HOBOE phase 3 randomised trial. European Journal of Cancer 2019;118:178-86. - PubMed
JONIE 2017 {published data only}
-
- Hasegawa Y, Kohno N, Horiguchi J, Miura D, Ishikawa T, Hayashi M, et al. A randomized controlled trial comparing zoledronic acid plus chemotherapy with chemotherapy alone as a neoadjuvant treatment in patients with HER2-negative primary breast cancer. Cancer Research 2012;72(24 Suppl):PD07-05.
-
- Horiguchi J, Hasegawa Y, Miura D, Ishikawa T, Hayashi M, Takao S, et al. A randomized controlled trial comparing zoledronic acid plus chemotherapy with chemotherapy alone as a neoadjuvant treatment in patients with HER2-negative primary breast cancer. Journal of Clinical Oncology 2013;31(15 Suppl):1029.
-
- Ishikawa T, Akazawa K, Hasegawa Y, Tanino H, Horiguchi J, Miura D, et al. Survival outcomes of neoadjuvant chemotherapy with zoledronic acid for HER2-negative breast cancer. Journal of Surgical Research 2017;220:46-51. - PubMed
-
- Ishikawa T, Akazawa K, Hasegawa Y, Tanino H, Horiguchi J, Miura D, et al. Zoledronic acid combined with neoadjuvant chemotherapy for HER2-negative early breast cancer (JONIE 1 trial): survival outcomes of a randomized multicenter phase 2 trial. Cancer Research 2017;77(4 Suppl 1):P5-16-10.
Kanis 1996 {published data only}
-
- Kanis JA, Powles T, Paterson AHG, McCloskey EV, Ashley S. Clodronate decreases the frequency of skeletal metastases in women with breast cancer. Bone 1996;19:663-7. - PubMed
-
- Powles TJ, Paterson A, Ashley S, Tidy A, McCloskey E, Nevantaus A, et al. Adjuvant clodronate will reduce the incidence of bone metastases in patients with primary operable breast cancer. Breast Cancer Research & Treatment 1998;50(3):234.
Kristensen 2008 {published data only}
-
- Kristensen B, Ejlertsen B, Mouridsen HT, Jensen MB, Andersen J, Bjerregaard B, et al. Bisphosphonate treatment in primary breast cancer: results from a randomised comparison of oral pamidronate versus no pamidronate in patients with primary breast cancer. Acta Oncologica (Stockholm, Sweden) 2008;47(4):740-6. - PubMed
Mardiak 2000 {published data only}
-
- Mardiak J, Bohunick L, Chovanec J, lek T, Koza I, Slovak Clodronate Collaborative Group. Adjuvant clodronate therapy in patients with locally advanced breast cancer - long term results of a double blind randomized trial.. Neoplasma 2000;47(3):177-80. - PubMed
Monda 2017 {published data only}
N02C1 2009 {published data only}
-
- Hines SL, Mincey BA, Sloan JA, Thomas SP, Chotinner EG, Loprinzi CL, et al. N02C1: a phase III randomized, placebo-controlled, double-blind trial of risedronate for prevention of bone loss in premenopausal women undergoing adjuvant chemotherapy for breast cancer (BC) [abstract no. 525]. Journal of Clinical Oncology 2008;26:12. - PMC - PubMed
-
- Hines SL, Mincey BA, Sloan JA, Thomas SP, Chottiner E, Loprinzi CL, et al. Phase III randomized, placebo-controlled, double-blind trial of risedronate for the prevention of bone loss in premenopausal women undergoing chemotherapy for primary breast cancer. Journal of Clinical Oncology 2009;27(7):1047-53. - PMC - PubMed
-
- NCT00054418. Risedronate in preventing bone loss in premenopausal women receiving chemotherapy for primary breast cancer. Https://clinicaltrials.gov/show/NCT00054418 (date received 6 February 2003).
NATAN 2016 {published data only}
-
- NCT00512993. Postoperative use of zoledronic acid in breast cancer patients after neoadjuvant chemotherapy. https://clinicaltrials.gov/ct2/show/NCT00512993 (date received 8 August 2007).
-
- Von Minckwitz G, Rezai M, Eidtmann H, Tesch H, Huober J, Gerber B, et al. Postneoadjuvant treatment with zoledronate in patients with tumor residuals after anthracyclines-taxane-based chemotherapy for primary breast cancer - the phase III NATAN study (GBG 36/ABCSG XX). Cancer Research 2013;73(24 Suppl):S5-05.
-
- Von Minckwitz G, Rezai M, Tesch H, Huober J, Gerber B, Zahm DM, et al. Zoledronate for patients with invasive residual disease after anthracyclines-taxane-based chemotherapy for early breast cancer - the Phase III NeoAdjuvant Trial Add-oN (NaTaN) study (GBG 36/ABCSG 29). European Journal of Cancer 2016;64:12-21. - PubMed
-
- Von Minckwitz G, Zahm MD, Eidtmann H, Tesch H, Du Bois A, Schwedler K, et al. Zoledronic acid (ZOL) as add-on therapy in patients with tumour residuals after neoadjuvant chemotherapy for primary breast cancer - first interim safety analysis of the NATAN study (GBG 36). European Journal of Cancer 2010;8(3 Suppl):65.
NEOZOL 2018 {published data only}
-
- Lelievre L, Clezardin P, Magaud L, Roche L, Tubiana-Mathieu N, Tigaud JD, et al. Comparative study of neoadjuvant chemotherapy with and without zometa for management of locally advanced breast cancer with serum VEGF as primary endpoint: the NEOZOL study. Clinical Breast Cancer 2018;18(6):e1311-21. - PubMed
-
- Mathevet P, Magaud L, Clezardin P. Adding zoledronic acid to neo-adjuvant chemotherapy may improve the efficiency of chemotherapy in locally advanced breast cancer: results from the prospective randomized study NEOZOL. Cancer Research 2016;76(4 Suppl):P6-13-19.
-
- NCT01367288. Comparative study of neoadjuvant chemotherapy with and without Zometa for management of locally advanced breast cancers. https://clinicaltrials.gov/show/NCT01367288 (date received 7 June 2010). - PubMed
NEO‐ZOTAC BOOG 2010 {published data only}
-
- Charehbili A, Hamdy NA, Smit VT, Kessels L, Van Bochove A, Van Laarhoven HW, et al. Vitamin d (25-0H D3) status and pathological response to neoadjuvant chemotherapy in stage II/III breast cancer: data from the NEOZOTAC trial (BOOG 10-01). Breast (Edinburgh, Scotland) 2016;25:69-74. - PubMed
-
- Charehbili A, Hamdy NAT, Smit VM, Liefers GJ, Putter H, Meershoek-Klein Kranenbarg E, et al. Changes in circulating vitamin D levels as a predictor for pathological response to neoadjuvant chemotherapy (NAC) in breast cancer (BC): a Dutch breast cancer trialists group (BOOG) side-study. Cancer Research 2013;73(24 Suppl):P1-08-19.
-
- Charehbili A, Van De Ven S, Liefers GJ, Smit VT, Wasser MN, Meershoek-Klein Kranenbarg EM, et al. Clinical and pathological response after neoadjuvant chemotherapy with or without zoledronic acid for patients with HER2-negative large resectable or stage II or III breast cancer. European Journal of Cancer 2013;49:S401.
-
- Charehbili A, Van De Ven S, Liefers GJ, Smit VTHBM, Putter H, Heijns JB, et al. NEOZOTAC: efficacy results from a phase III randomized trial with neoadjuvant chemotherapy (TAC) with or without zoledronic acid for patients with HER2-negative large resectable or stage II or III breast cancer (BC)-a Dutch Breast Cancer Trialists' Group (BOOG) study. Journal of Clinical Oncology 2013;31(15 Suppl):1028.
-
- Charehbili A, Van de Ven, Smit VT, Meershoek-Klein Kranenbarg E, Hamdy NA, Putter H, et al. Addition of zoledronic acid to neoadjuvant chemotherapy does not enhance tumor response in patients with HER2-negative stage II/III breast cancer: the NEOZOTAC trial (BOOG 2010-01). Annals of Oncology 2014;25(5):998-1004. - PubMed
Novartis I 2006 {published data only}
-
- NCT00332709. Safety/efficacy of letrozole monotherapy or in combination with zoledronic acid as extended adjuvant treatment of postmenopausal patients with primary breast cancer. https://clinicaltrials.gov/ct2/show/NCT00332709 (date received 2 June 2006).
NSABP B‐34 2012 {published data only}CDR0000068426
-
- NCT00009945. Clodronate with or without chemotherapy and/or hormonal therapy in treating women with stage I or stage II breast cancer. https://clinicaltrials.gov/ct2/show/NCT00009945 (date received 27 January 2003).
-
- Paterson AH, Anderson SJ, Lembersky BC, Fehrenbacher L, Falkson CI, King KM, et al. Oral clodronate for adjuvant treatment of operable breast cancer (National Surgical Adjuvant Breast and Bowel Project protocol B-34): a multicentre, placebo-controlled, randomised trial. Lancet. Oncology 2012;13(7):734-42. - PMC - PubMed
-
- Paterson AHG, Anderson SJ, Lembersky BC, Fehrenbacher L, Falkson CI, King KM, et al. NSABP Protocol B-34: a clinical trial comparing adjuvant clodronate vs. placebo in early stage breast cancer patients receiving systemic chemotherapy and/or tamoxifen or no therapy - final analysis 6. Cancer Research 2011;71(24 Suppl):S2-3.
-
- Paterson AHG, Anderson SJ, Lembersky BC, Fehrenbacher L, Falkson CI, King KM, et al. NSABP protocol B-34: a clinical trial comparing adjuvant clodronate vs. placebo in early stage breast cancer patients receiving systemic chemotherapy and/or tamoxifen or no therapy - final analysis. Cancer Research 2011;71(24 Suppl):S2-3.
Powles 2006 {published data only}ISRCTN83688026
-
- Atula S, Powles T, Paterson A, McCloskey E, Nevalainen J, Kanis J. Extended safety profile of oral clodronate after long-term use in primary breast cancer patients. Drug Safety 2003;26(9):661-71. - PubMed
-
- Atula ST, Paterson AHG, Powles TJ, McCloskey EV, Nevalamen JI, Kants JA. Safety profile of oral clodronate during long-term use in primary breast cancer patients. Bone 2002;30(3):46S. - PubMed
-
- ISRCTN83688026. A randomised controlled clinical trial to evaluate the anti-osteolytic agent clodronate for the prevention of the development of bone metastases in patients with primary breast cancer. https://www.isrctn.com/ISRCTN83688026 (date received 1 February 2006).
-
- McCloskey E, Paterson A, Kanis J, Tahtela R, Powles T. Effect of oral clodronate on bone mass, bone turnover and subsequent metastases in women with primary breast cancer. European Journal of Cancer 2010;46(3):558-65. - PubMed
-
- McCloskey E, Paterson AH, Powles TJ. Oral clodronate maintains bone mass in women with primary breast cancer. Journal of Clinical Oncology 2005;23(16 Suppl):12S.
ProBONE I 2005 {published data only}
-
- EUCTR2004-002831-14-DE. Influence of zoledronic acid (Zometa®) on bone mineral density and bone ultrasonometry in premenopausal women with hormone receptor negative breast cancer and adjuvant chemotherapeutic treatment. https://www.clinicaltrialsregister.eu/ctr-search/trial/2004-002831-14/DE (date received 2005).
-
- Hadji P, Kauka A, Bauer T, Kalder M, Albert U, Birkholz K, et al. Influence of zoledronic acid on BMD in premenopausal women with breast cancer and neoadjuvant or adjuvant chemotherapy and/or endocrine treatment - the Probone studies. Cancer Research 2011;71(24 Suppl):P2-19-03.
ProBONE II 2015 {published data only}
-
- Hadji P, Kauka A, Bauer T, Kalder M, Albert U, Birkholz K, et al. Influence of zoledronic acid on BMD in premenopausal women with breast cancer and neoadjuvant or adjuvant chemotherapy and/or endocrine treatment - the Probone studies. Cancer Research 2011;71(24 Suppl):P2-19-03.
-
- Hadji P, Kauka A, Bauer T, Kalder M, Albert US, Birkholz K, et al. The ProBone study: influence of zoledronic acid on bone mineral density in premenopausal women with breast cancer and neoadjuvant or adjuvant chemotherapy and/or endocrine treatment. Journal of Cancer Research and Clinical Oncology 2012;138:62.
-
- Hadji P, Kauka A, Kalder M, Bauer T, Albert U, Muth M, et al. Influence of zoledronic acid on bone mineral density in premenopausal women with hormone receptor positive or negative breast cancer and neoadjuvant or adjuvant chemotherapy or endocrine treatment. European Journal of Cancer 2010;8(3 Suppl):62.
-
- Hadji P, Kauka A, Ziller M, Birkholz K, Baier M, Muth M, et al. Effect of adjuvant endocrine therapy on hormonal levels in premenopausal women with breast cancer: the ProBONE II study. Breast Cancer Research & Treatment 2014;144(2):343-51. - PubMed
-
- Hadji P, Kauka A, Ziller M, Birkholz K, Baier M, Muth M, et al. Effects of zoledronic acid on bone mineral density in premenopausal women receiving neoadjuvant or adjuvant therapies for HR+ breast cancer: the ProBONE II study. Osteoporosis International 2014;25(4):1369-78. - PubMed
REBBeCA 2008 {published data only}
-
- Greenspan SL, Bhattacharya RK, Sereika SM, Brufsky A, Vogel VG. Prevention of bone loss in survivors of breast cancer: a randomized, double-blind, placebo-controlled clinical trial. Journal of Clinical Endocrinology and Metabolism 2007;92(1):131-6. - PubMed
-
- NCT00118508. The prevention of osteoporosis in premenopausal and newly postmenopausal (up to 8 years) women with breast cancer following chemotherapy (REBBeCA study). https://www.clinicaltrials.gov/ct2/show/NCT00118508 (date received 11 July 2005).
REBBeCA II 2016 {published data only}
-
- Anonymous. Once-weekly risedronate benefits postmenopausal breast-cancer survivors. Nature Clinical Practice Endocrinology and Metabolism 2008;4:478.
-
- Greenspan S, Perera S, Vujevich K, Van Londen G, Brufsky A, Lembersky B, et al. Prevention of bone loss in breast cancer survivors on aromatase inhibitors: results of the Rebbeca II trial. Journal of Bone and Mineral Research (conference abstracts) 2013;28:S18.
-
- NCT00485953. Effect of bisphosphonate on bone loss in postmenopausal women with breast cancer initiating aromatase inhibitor therapy. Https://clinicaltrials.gov/show/NCT00485953 (date received 13 June 2007).
Rhee 2013 {published data only}
-
- Rhee Y, Song K, Lim SK, Park BW. Efficacy of a combined alendronate and calcitriol agent in Korean postmenopausal women with early breast cancer receiving aromatase inhibitor: a double-blind, randomized, placebo-controlled study. Osteoporosis International 2010;5:S760-1. - PubMed
-
- Rhee Y, Song K, Park S, Park HS, Lim SK, Park BW. Efficacy of a combined alendronate and calcitriol agent (maxmarvil) in Korean postmenopausal women with early breast cancer receiving aromatase inhibitor: a double-blind, randomized, placebo-controlled study. Endocrine Journal 2013;60(2):167-72. - PubMed
Saarto 2004 {published data only}
-
- Leppa S, Saarto T, Vehmanen L, Blomqvist C, Elomaa I. Clodronate treatment influences MMP-2 associated outcome in node positive breast cancer. Breast Cancer Research & Treatment 2005;90(2):117-25. - PubMed
-
- Saarto T, Blomqvist C, Valimaki M, Makela P, Elomaa I. Clodronate improves bone mineral density in early breast cancer patients. A randomized study [abstract no: 51]. European Journal of Cancer 1995;31A(Suppl 5):S12.
-
- Saarto T, Blomqvist C, Valimaki M, Makela P, Sarna S, Elomaa I. Chemical castration induced by adjuvant cyclophosphamide, methotrexate, and fluorouracil chemotherapy causes rapid bone loss that is reduced by clodronate: a randomized study in premenopausal breast cancer patients. Journal of Clinical Oncology 1997;15(4):1341-7. - PubMed
SABRE 2010 {published data only}
-
- NCT00082277. Anastrozole biphosphonate study in postmenopausal women with hormone-receptor-positive early breast cancer. https://clinicaltrials.gov/ct2/show/NCT00082277 (date received 6 May 2004).
-
- Van Poznak C, Hannon R, Clack G, Campone M, Mackey JR, Apffelstaedt J, et al. Managing cancer treatment-induced bone loss: 24-month results from the Study of Anastrozole with the Bisphosphonate Risedronate. Cancer Research 2009;69(2 Suppl):1137.
-
- Van Poznak C, Hannon R, Clack G, Campone M, Mackey JR, Apffelstaedt J, et al. Managing cancer treatment-induced bone loss: 24-month results from the study of anastrozole with the bisphosphonate risedronate (SABRE). Cancer Research 2009;69(2 Suppl):1137.
-
- Van Poznak C, Hannon RA, Clack G, Campone M, Mackey JR, Apffelstaedt J, et al. The SABRE (Study of Anastrozole with the Bisphosphonate RisedronatE) study: 12-month analysis. Breast Cancer Research and Treatment 2007;106:S37.
-
- Van Poznak C, Hannon RA, Mackey JR, Campone M, Apffelstaedt JP, Clack G, et al. Prevention of aromatase inhibitor-induced bone loss using risedronate: the SABRE trial. Journal of Clinical Oncology 2010;28(6):967-75. - PubMed
Safra 2011 {published data only}
-
- Greenberg J, Stemmer SM, Bernstein-Molho R, Pelles-Avraham S, Stephansky I, Inbar MJ, et al. The protective effect of zoledronic acid on bone loss in postmenopausal women with early breast cancer treated with sequential tamoxifen and letrozole: 36-month follow-up. Journal of Clinical Oncology 2011;29(15 Suppl 1):e11111. - PubMed
-
- NCT00376740. Effectiveness of zoledronic acid in the prevention of osteoporosis in early breast cancer patients receiving letrozole. https://clinicaltrials.gov/ct2/show/NCT00376740 (date received 15 September 2006).
-
- Safra T, Bernstein Molho R, Stephansky I, Yaal-Hahoshen N, Inbar M, Ackerstein A, et al. Effect of zoledronic acid on bone loss in postmenopausal women with early breast cancer treated with sequential tamoxifen and letrozole. Journal of Clinical Oncology 2009;27(15S):599. - PubMed
-
- Safra T, Bernstein-Molho R, Greenberg J, Pelles-Avraham S, Stephansky I, Sarid D, et al. The protective effect of zoledronic acid on bone loss in postmenopausal women with early breast cancer treated with sequential tamoxifen and letrozole: a prospective, randomized, phase II trial. Oncology 2011;81(5-6):298-305. - PubMed
Saito 2015 {published data only}
-
- Saito M, Matsuoka J. Open-label randomized parallel controlled study comparing bone mineral density between alendronate plus alfacalcidol combination and single administration of alfacalcidol in postmenopausal women receiving aromatase inhibitor as adjuvant therapy. Cancer Research 2015;75(9 Suppl):P5-15-06.
Solomayer 2012 {published data only}
-
- NCT00172068. Zoledronic acid in the treatment of breast cancer with minimal residual disease in the bone marrow. https://www.clinicaltrials.gov/ct2/show/NCT00172068 (date received 15 September 2005).
-
- Solomayer E, Gebauer G, Hirnle P, Janni W, Lück H, Becker S, et al. Influence of zoledronic acid on disseminated tumor cells (DTC) in primary breast cancer patients. Cancer Research 2009;69(2 Suppl):2048. - PubMed
-
- Solomayer EF, Banys M, Krawczyk N, Gebauer G, Wallwiener D, Hirnle P, et al. Bisphosphonates may improve survival of breast cancer patients with disseminated tumor cells in bone marrow. Journal of Cancer Research and Clinical Oncology 2012;1:90.
-
- Solomayer EF, Gebauer G, Hirnle P, Janni W, Luck HJ, Becker S, et al. Influence of zoledronic acid on disseminated tumor cells in primary breast cancer patients. Annals of Oncology 2012;23(9):2271-7. - PubMed
Sun 2016 {published data only}
SWOG S0307 2019 {published data only}
-
- Gralow J, Barlow WE, Paterson AHG, Lew D, Stopeck A, Hayes DF, et al. Phase III trial of bisphosphonates as adjuvant therapy in primary breast cancer: sWOG/Alliance/ECOG-ACRIN/NCIC Clinical Trials Group/NRG Oncology study S0307. Journal of Clinical Oncology 2015;33(15 Suppl):503.
-
- Gralow J, Barlow WE, Paterson AHG, Lew D, Stopeck A, Hayes DF, et al. SWOG S0307 phase III trial of bisphosphonates as adjuvant therapy in primary breast cancer: comparison of toxicities and patient-stated preference for oral versus intravenous delivery. Journal of Clinical Oncology 2014;32(15 Suppl):558.
-
- Kizub D, Miao J, Stopeck A, Thompson P, Paterson AH, Clemons M, et al. Statin use, site of recurrence, and survival among post-menopausal women taking bisphosphonates as adjuvant therapy for breast cancer (SWOG S0307). Cancer Research 2019;79(4 Suppl):P1-17-03.
-
- Kizub DA, Miao J, Schubert MM, Paterson AHG, Clemons M, Dees EC, et al. Factors associated with osteonecrosis of the jaw in women with breast cancer receiving high-dose bisphosphonates to prevent breast cancer metastases as part of the SWOG 0307 trial. Journal of Clinical Oncology 2019;37(Suppl 15):552.
Team IIB 2006 {published data only}
-
- ISRCTN17633610. TEAM II: a randomised, multicentre, prospective, phase III trial investigating neoadjuvant hormonal therapy with exemestane for three versus six months (TEAM IIa) and/or the efficacy and safety of the addition of ibandronate to adjuvant hormonal therapy in post-menopausal women with hormone receptor positive early breast cancer (TEAM IIb). http://www.who.int/trialsearch/Trial2.aspx?TrialID=ISRCTN17633610 (date received 28 December 2006).
-
- NTR785. TEAM II A randomised, multicentre, prospective, phase III trial investigating TEAM IIa: neoadjuvant hormonal therapy with exemestane for three versus six months. and / or TEAM IIb: the efficacy and safety of the addition of ibandronate to adjuvant hormonal therapy in postmenopausal women with hormone receptor positive early breast cancer. http://www.who.int/trialsearch/Trial2.aspx?TrialID=NTR785 (date received 2006).
-
- Vliek SB, Meershoek-Klein Kranenbarg E, Van Rossum AG, Tanis BC, Putter H, Van Der Velden AW, et al. The efficacy and safety of the addition of ibandronate to adjuvant hormonal therapy in postmenopausal women with hormone-receptor positive early breast cancer. First results of the TEAM IIB trial (BOOG 2006-04). Cancer Research 2017;77(4 Suppl 1):S6-02.
-
- Vliek SB, Noordhoek I, Meershoek-Klein Kranenbarg E, Van Rossum AGJ, Dezentje VO, Jager A, et al. Daily oral ibandronate with adjuvant endocrine therapy in postmenopausal women with estrogen receptor-positive breast cancer (BOOG 2006-04): randomized phase III TEAM-IIB trial. Journal of Clinical Oncology 2022;40(25):2934-45 Erratum in: Journal of Clinical Oncology 2022; 40(28):3352. - PubMed
Tevaarwerk 2007 {published data only}
-
- NCT00213980. Bone mineral density effects of zoledronate in postmenopausal women with breast cancer. https://clinicaltrials.gov/show/NCT00213980 (date received 21 September 2005).
-
- Tevaarwerk A, Stewart JA, Love R, Binkley NC, Black S, Eickhoff J, et al. Randomized trial to assess bone mineral density (BMD) effects of zoledronic acid (ZA) in postmenopausal women (PmW) with breast cancer. Journal of Clinical Oncology 2007;25(18 Suppl):19558.
References to studies excluded from this review
Ahmad 2007 {published data only}
-
- Ahmad K. Zoledronic acid prevents bone loss. Lancet Oncology 2007;8(5):375. - PubMed
Ahn 2009 {published data only}
-
- Ahn J, Jung K, Kim S, Lee K, Ro J, Park Y, et al. Zoledronic acid prevents bone loss in premenopausal women with early breast cancer undergoing adjuvant chemotherapy: a phase III study of Korean Cancer Study Group (KCSG-BR06-01). Cancer Research 2009;69(24 Suppl):2104. - PubMed
-
- Kim JE, Ahn JH, Jung KH, Kim SB, Kim HJ, Lee KS, et al. Zoledronic acid prevents bone loss in premenopausal women with early breast cancer undergoing adjuvant chemotherapy: a phase III trial of the Korean Cancer Study Group (KCSG-BR06-01). Breast Cancer Research and Treatment 2011;125(1):99-106. - PubMed
BATMAN 2005 {published data only}
-
- NCT00122356. Bisphosphonate and anastrozole trial - bone maintenance algorithm assessment. https://clinicaltrials.gov/ct2/show/NCT00122356 (date received 22 July 2005).
CALGB 79809 {published data only}
-
- Anonymous. CALGB 78909: phase III trial of intravenous zoledronic acid in the prevention of bone loss in localized breast cancer patients with chemotherapy-induced ovarian failure. Clinical Advances in Hematology & Oncology 2005;3:105-6. - PubMed
-
- NCT00022087. Zoledronate, calcium, and vitamin D in preventing bone loss in women receiving adjuvant chemotherapy for breast cancer. Https://clinicaltrials.gov/show/NCT00022087 (date received 27 January 2003).
-
- Shapiro CL, Halabi S, Gibson G, Weckstein DJ, Kirshner J, Sikov WM, et al. Effect of zoledronic acid (ZA) on bone mineral density (BMD) in premenopausal women who develop ovarian failure (OF) due to adjuvant chemotherapy (AdC): first results from CALGB trial 7980. Journal of Clinical Oncology 2008;26(15 Suppl):512.
Chen 2011 {published data only}
-
- Chen J, Liu ZY, Zhao L. Effects of zoledronic acid in the treatment of breast cancer. Chung-hua Chung Liu Tsa Chih [Chinese Journal of Oncology] 2011;33:318-9. - PubMed
Ciardo 2020 {published data only}
-
- Ciardo D, Casciaro E, Ciccarese M, Conversano F, Forcignano R, Lombardi F, et al. Rems technology for short-term monitoring of denosumab therapeutic effect in breast cancer patients receiving aromatase inhibitors based therapy. Osteoporosis International 2020;31(Suppl 1):S372-3.
Fuleihan 2005 {published data only}
-
- Fuleihan G, Salamoun M, Mourad YA, Chehal A, Salem Z, Mahfoud Z, et al. Pamidronate in the prevention of chemotherapy-induced bone loss in premenopausal women with breast cancer: a randomized controlled trial. Journal of Clinical Endocrinology & Metabolism 2005;90(6):3209-14. - PubMed
Gessner 2000 {published data only}
-
- Gessner U, Koeberle D, Thuerlimann B, Bacchus L, Horisberger B. Economic analysis of terminal care for patients with malignant osteolytic bone disease and pain treated with pamidronate. Supportive Care in Cancer 2000;8(2):115-22. - PubMed
Gucalp 1994 {published data only}
-
- Gucalp R, Theriault R, Gill I, Madajewicz S, Chapman R, Navari R, et al. Treatment of cancer-associated hypercalcemia. Double-blind comparison of rapid and slow intravenous infusion regimens of pamidronate disodium and saline alone. Archives of Internal Medicine 1994;154(17):1935-44. - PubMed
Hines 2010 {published data only}
-
- Majithia N, Atherton PJ, Lafky JM, Wagner-Johnston N, Olson J, Dakhil SR, et al. Zoledronic acid for treatment of osteopenia and osteoporosis in women with primary breast cancer undergoing adjuvant aromatase inhibitor therapy: a 5-year follow-up. Supportive Care in Cancer 2016;24(3):1219-26. - PubMed
IBIS 3 FEASIBILITY {published data only}ISRCTN93764730
-
- EUCTR2014-004430-26-GB. Feasibility of the International Breast Intervention Study 3 (IBIS 3). Prevention of late recurrence in hormone receptor positive breast cancer survivors following 5 years of treatment. http://www.who.int/trialsearch/Trial2.aspx?TrialID=EUCTR2014-004430-26-GB (date received 2015).
-
- ISRCTN93764730. Feasability of IBIS 3. An international breast intervention study investigating prevention of late recurrence in ER+ breast cancer survivors following 5 years of adjuvant treatment. https://www.clinicaltrialsregister.eu/ctr-search/trial/2014-004430-26/re... (date received 2014).
IBIS II 2003 {published data only}
-
- Sestak I, Cuzick J, Blake G, Patel R, Coleman R, Eastell R. Effect of risedronate on bone loss due to anastrozole given to prevent breast cancer: 5-year results from the IBISII prevention trial. Journal of Bone and Mineral Research 2016;31(Suppl 1):S102.
-
- Sestak I, Cuzick J. Off-treatment bone mineral density changes in postmenopausal women after 5 years of anastrozole. Journal of Bone and Mineral Research 2018;33(Suppl 1):102-3.
-
- Singh S, Cuzick J, Blake GM, Mesher D, Patel R, Truscott J, et al. One year effect of anastrozole and risedronate on bone mineral density: first results from the IBIS-II bone sub-study. Bone 2011;48(1):S24.
JPRN‐UMIN000004375 {published data only}
-
- JPRN-UMIN000004375. Study on preventive effect of risedronate administration on aromatase inhibitor-induced bone mineral loss in the endoclinical therapy for postmenopausal breast cancer patients. http://www.who.int/trialsearch/Trial2.aspx?TrialID=JPRN-UMIN000004375 (date received 2010).
Lee 2011 {published data only}
-
- Lee SA, Hwang SH, Ahn SG, Lee HM, Jeong J, Lee H. Effects of zoledronic acid on bone mineral density during aromatase inhibitor treatment of Korean postmenopausal breast cancer patients. Breast Cancer Research and Treatment 2011;130:863-70. - PubMed
Lipton 1999 {published data only}
-
- Lipton A, Berenson J, Knight R, Grace Hu, Levvy E. Phase 2 trial of zoledronate vs pamidronate in multiple myeloma and breast cancer. In: European Cancer Conference 10, Vienna. Vol. 35 Suppl 4. 1999:S360.
N03CC {published data only}
-
- Hines SL, Mincey BA, Sloan JA, Thomas SP, Chottiner E, Loprinzi CL, et al. Phase III randomized, placebo-controlled, double-blind trial of risedronate for the prevention of bone loss in premenopausal women undergoing chemotherapy for primary breast cancer. Journal of Clinical Oncology 2009;27(7):1047-53. - PMC - PubMed
-
- Mincey BA, Dentchev T, Sloan JA, Hines SL, Perez EA, Johnson DB, et al. N03CC - a randomized, controlled, open-label trial of upfront vs. delayed zoledronic acid for prevention of bone loss in postmenopausal (PM) women with primary breast cancer (PBC) starting letrozole after tamoxifen. Journal of Clinical Oncology 2008;26(15 Suppl):564.
-
- NCT00107263. Zoledronate in preventing bone loss in postmenopausal women who are receiving letrozole for stage I, stage II, or stage IIIA breast cancer. https://clinicaltrials.gov/show/NCT00107263 (date received 6 April 2005).
Nakatsukasa 2019 {published data only}
-
- Nakatsukasa K, Koyama H, Ouchi Y, Ono H, Sakaguchi K, Matsuda T, et al. Effect of denosumab on low bone mineral density in postmenopausal Japanese women receiving adjuvant aromatase inhibitors for non-metastatic breast cancer: 24-month results. Breast Cancer (Tokyo, Japan) 2019;26(1):106-12. - PubMed
NCT00196859 {published data only}
-
- NCT00196859. Study in elderly patients with early breast cancer (ICE). https://clinicaltrials.gov/show/NCT00196859 (date received 20 September 2005).
NCT00202059 {published data only}
-
- NCT00202059. Effects of zometa and physical activity on bone density in women receiving chemotherapy for breast cancer. https://clinicaltrials.gov/ct2/show/NCT00202059 (date received 20 September 2005).
NCT00247650 {published data only}
-
- NCT00247650. Comparison study of letrozole alone or letrozole with zoledronic acid in early breast cancer, neoadjuvant therapy. https://www.clinicaltrials.gov/ct2/show/NCT00247650 (date received 1 November 2005).
NCT00295867 {published data only}
-
- NCT00295867. Zoledronate in treating bone marrow micrometastases in women with stage I, stage II, or stage III breast cancer. https://www.clinicaltrials.gov/ct2/show/NCT00295867 (date received 24 February 2006).
NCT00324714 {published data only}
-
- NCT00324714. Risedronate in improving bone mineral density and bone health in postmenopausal women with ductal carcinoma in situ enrolled in clinical trial CRUK-IBIS-II-DCIS. https://clinicaltrials.gov/ct2/show/NCT00324714 (date received 11 May 2006).
NCT00873808 {published data only}
-
- NCT00873808. S0307A, long-term bone quality in women with breast cancer enrolled on clinical trial SWOG-S0307. https://clinicaltrials.gov/ct2/show/NCT00873808 (date received 2 April 2009).
NCT02051218 {published data only}
-
- NCT02051218. Prevention of symptomatic skeletal events with denosumab administered every 4 weeks versus every 12 weeks. https://clinicaltrials.gov/ct2/show/NCT02051218 (date received 31 January 2014).
NCT03358017 {published data only}
-
- NCT03358017. Neoadjuvant zoledronate and atorvastatin in triple negative breast cancer (YAPPETIZER). https://clinicaltrials.gov/ct2/show/NCT03358017 (date received 30 November 2017).
NCT03664687 {published data only}
-
- NCT03664687. Comparing a single-dose vs. twice yearly zoledronate in patients with early stage breast cancer (REaCT-ZOL). https://clinicaltrials.gov/ct2/show/NCT03664687 (date received 10 September 2018).
NCT05164952 {published data only}
-
- NCT05164952. Delayed versus immediate use of zoledronic acid for postmenopausal patients with ER/PR positive early breast cancer who are using adjuvant letrozole. https://clinicaltrials.gov/ct2/show/NCT05164952 (date received 21 December 2021).
PERIDENO {published data only}
-
- EUCTR2016-005210-22-NL. The influence of denosumab on the immune system in women after the menopause with HER2 negative breast cancer. https://www.clinicaltrialsregister.eu/ctr-search/trial/2016-005210-22/NL (date received 2017).
-
- NCT03532087. Study to identify the impact of denosumab on the immune system in patients with HER2 negative breast cancer. Https://clinicaltrials.gov/show/NCT03532087 (date received 22 May 2018).
Purohit 1995 {published data only}
Ralston 1997 {published data only}
Rizzoli 1996 {published data only}
-
- Rizzoli R, Forni M, Schaad MA, Slosman DO, Sappino AP, Garcia J, et al. Effects of oral clodronate on bone mineral density in patients with relapsing breast cancer. Bone 1996;18(6):531-7. - PubMed
Smith 1999 {published data only}
-
- Smith TJ, Hillner BE. Clodronate reduced the incidence of bony and visceral metastases in patients with breast cancer and tumour cells in the bone marrow. Evidence Based Medicine 1999;4(2):43.
SUCCESS {published data only}
-
- Andergassen U, Rack B, Schindlbeck C, Raber G, Ulmer H, Heinrich G, et al. Evaluation of CA 27.29 as prognostic marker in primary breast cancer patients - results of the German SUCCESS trial. Cancer Research 2009;69(24 Suppl):3023.
-
- Bauer ECA, Neugebauer JK, Andergassen U, Jaeger B, Jueckstock JK, Fasching PA, et al. Evaluation of prevalence, number, and temporal changes of circulating tumor cells as assessed after 2 and 5 years of follow-up in patients with early breast cancer in the SUCCESS: a study. Journal of Clinical Oncology 2013;31(15 Suppl):11042.
-
- Bauer ECA, Schochter F, Widschwendter P, DeGregorio A, Andergassen U, Friedl TWP, et al. Prevalence of circulating tumor cells in early breast cancer patients 2 and 5 years after adjuvant treatment. Breast Cancer Research & Treatment 2018;171(3):571-80. - PubMed
-
- De Gregorio A, Rack B, Fasching PA, Haberle L, Friedl TWP, Tesch H, et al. Persistence of circulating tumor cells in high-risk early breast cancer patients during follow-up care suggests poor prognosis: results from the adjuvant SUCCESS A trial. Clinical and Experimental Metastasis 2018;35(3):202.
Takahashi 2012 {published data only}
-
- Kohno N, Hasegawa Y, Horiguchi J, Ishikawa T, Miura D, Hayashi M, et al. Randomized controlled trial comparing zoledronic acid plus chemotherapy with chemotherapy alone as a neoadjuvant treatment in patients with HER2-negative primary breast cancer. Journal of Clinical Oncology (conference abstracts) 2012;30(15 Suppl):TPS1141.
-
- Takahashi S, Iwase T, Kohno N, Ishikawa T, Kim SJ, Hosoda M, et al. Efficacy of zoledronic acid in postmenopausal Japanese women with early breast cancer receiving adjuvant letrozole: 36-month results. Journal of Clinical Oncology 2013;31(15 Suppl):e20510.
-
- Takahashi S, Iwase T, Kohno N, Ishikawa T, Taguchi T, Takahashi M, et al. Efficacy of zoledronic acid in postmenopausal Japanese women with early breast cancer receiving adjuvant letrozole: 12-month results. Breast Cancer Research & Treatment 2012;133(2):685-93. - PubMed
-
- Takahashi S, Iwase T, Kohno N, Ishikawa T, Taguchi T, Takahashi M, et al. Zoledronic acid inhibits adjuvant letrozole-associated bone loss in postmenopausal Japanese women with early breast cancer. Journal of Clinical Oncology 2011;29:829-36.
-
- UMIN-CTR700207118. Assessment of the efficacy of the use of zoledronic acid in the prevention of aromatase inhibitor-associated bone loss in postmenopausal women with hormone receptor-positive breast cancer who received letrozole as adjuvant therapy. https://adisinsight.springer.com/trials/700207118 (date received 2008).
Toulis 2016 {published data only}
-
- Toulis K, Iliadou P, Mandanas S, Kazila P, Margaritidou E, Georgopoulos K, et al. Calcium homeostasis in women with non-metastatic breast cancer with osteoporosis after a single dose of denosumab: a pilot study. Hormones 2016;15:560-2. - PubMed
Van Hellemond 2019 {published data only}
Vinholes 1995 {published data only}
-
- Vinholes J, Purohit OP, Guo CY, Eastell R, Vinholes AP, Coleman R. Randomised double-blind comparison of pamidronate or clodronate for hypercalcaemia of malignancy: effects on bone metabolism markers. European Journal of Cancer 1995;31A(Suppl 5):S253.
Vriens 2017 {published data only}
Z‐FAST 2012 {published data only}
-
- Brufsky A, Bosserman L, Caradonna R, Haley B, Jones M, Moore H, et al. The effect of zoledronic acid on aromatase inhibitor associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: the Z-FAST study 36-month follow-up. Clinical Breast Cancer 2009;29(2):77-85. - PubMed
-
- Brufsky A, Bosserman L, Caradonna R, Haley B, Jones M, Moore H, et al. The effect of zoledronic acid on aromatase inhibitor associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: the Z-FAST study 36-month follow-up. In: 30th Annual San Antonio Breast Cancer Symposium. 2007. - PubMed
-
- Brufsky A, Harker G, Beck J, Carroll R, Jin L, Warsi G, et al. The effect of zoledronic acid on aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: the Z-FAST study 5-year final follow-up. Cancer Research 2009;69(24 Suppl):4083.
-
- Brufsky A, Harker WG, Beck JT, Carroll R, Tan-Chiu E, Seidler C, et al. Zoledronic acid (ZA) effectively inhibits cancer treatment-induced bone loss (CTIBL) in postmenopausal women (PMW) with early breast cancer (BCa) receiving adjuvant letrozole (Let): 12 mos BMD resluts of the Z-FAST trial. Journal of Clinical Oncology 2005;23(16 Suppl):12S.
-
- Brufsky A, Harker WG, Beck JT, Carroll R, Tan-Chiu E, Seidler C, et al. Zoledronic acid inhibits adjuvant letrozole-induced bone loss in postmenopausal women with early breast cancer. Journal of Clinical Oncology 2007;25(7):829-36. - PubMed
ZO‐FAST 2013 {published data only}
-
- Llombart A, Frassoldati A, Paija O, Sleeboom HP, Jerusalem G, Mebis J, et al. Immediate administration of zoledronic acid reduces aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer: 12-month analysis of the E-ZO-FAST trial. Clinical Breast Cancer 2012;12(1):40-8. - PubMed
-
- Bundred NJ, Campbell ID, Davidson N, DeBoer RH, Eidtmann H, Monnier A, et al. Effective inhibition of aromatase inhibitor-associated bone loss by zoledronic acid in postmenopausal women with early breast cancer receiving adjuvant letrozole: ZO-FAST study results. Cancer 2008;112(5):1001-10. - PubMed
-
- Coleman R, De Boer R, Eidtmann H, Llombart A, Davidson N, Neven P, et al. Zoledronic acid (zoledronate) for postmenopausal women with early breast cancer receiving adjuvant letrozole (ZO-FAST study): final 60-month results. Annals of Oncology 2013;24(2):398-405. - PubMed
-
- Coleman R, De Boer R, Eidtmann H, Neven P, Von Minckwitz G, Martin N, et al. Influence of delayed zoledronic acid initiation on disease-free survival in postmenopausal women with endocrine receptor-positive early breast cancer receiving adjuvant letrozole: exploratory analyses from the ZO-FAST trial. Cancer Research 2011;71(24 Suppl):P2-17-01.
-
- De Boer R, Bundred N, Eidtmann H, Neven P, Von Minckwitz G, Martin N, et al. Long-term survival outcomes among postmenopausal women with hormone receptor-positive early breast cancer receiving adjuvant letrozole and zoledronic acid: 5-year follow-up of ZO-FAST. Cancer Research 2011;71(24 Suppl):S1-3.
ZOLMENO 2017 {published data only}
-
- ISRCTN15749696. Does zoledronic acid alter levels of reproductive hormones and how does this affect the tumour and bone in pre- and post-menopausal women with early breast cancer? https://www.isrctn.com/ISRCTN15749696 (date received 3 October 2017).
-
- Theodoulou E, Wilson C, Brown JE, Holen I. The ZOLMENO study-exploring the effects of ZOLedronic Acid and MENOpausal status in patients with early breast cancer. Journal of Bone and Mineral Research Plus 2019;3(Suppl 3):62-3. [ISRCTN: 15749696]
References to studies awaiting assessment
ACTRN12616001051437 {published data only}
-
- ACTRN12616001051437. A randomised, double-blind, placebo controlled trial to examine the effects of total oestradiol depletion on bone microstructure and the efficacy of denosumab in preventing microstructural bone decay in premenopausal women with early breast cancer. https://anzctr.org.au/Trial/Registration/TrialReview.aspx?ACTRN=12616001... (date received 5 August 2016).
-
- Ramchand SK, Cheung Y, Yeo B, Grossmann M. The effects of adjuvant endocrine therapy on bone health in women with breast cancer. Journal of Endocrinology 2019;241(3):R111. - PubMed
ADAIDO {published data only}
-
- EUCTR2020-005343-23-IT. Study of the antiresorptive treatment with alendronate versus no treatment after denosumab and aromatase inhibitors discontinuation in low fracture risk osteopenic postmenopausal women with non metastatic hormonal receptor positive breast cancer. https://www.clinicaltrialsregister.eu/ctr-search/trial/2020-005343-23/IT (date received 2021).
ChiCTR‐TRC‐09000408 {published data only}
-
- ChiCTR-TRC-09000408. A clinical study of the effects of zoledronic acid in the prevention of metastasis of breast cancer to bone, and impact for disease process of breast cancer patients. https://www.chictr.org.cn/showprojEN.html?proj=9125 (date received 27 April 2009).
El‐Ibrashi 2016 {published data only}
-
- El-Ibrashi MM, El-Sadda WM, Abdel-Halim II, Elashri MS. Zoledronic acid combined with adjuvant tamoxifen with or without ovarian function suppression in premenopausal early breast cancer patients. Cancer Research 2016;76(4 Suppl):P5-15-04.
Gunmalm 2018 {published data only}
-
- Gunmalm V, Rasmussen AQ, Lund-Jacobson T, Andersson M, Brons C, Schwarz P. Bone and calcium metabolic changes during anti-resorptive treatment in early breast cancer. Calcified Tissue International 2018;102(1 Suppl 1):75.
NCT02595138 {published data only}
-
- NCT02595138. Zoledronic acid as adjuvant treatment of triple-negative breast cancer. https://clinicaltrials.gov/show/NCT02595138 (date received 3 November 2015).
Rhee 2011 {published data only}
-
- Rhee Y, Jung Chung Y, Kim SH, Lim SK, Park BW. Aromatase inhibitor significantly increased circulating sclerostin level in patients with endocrine-responsive breast cancer. Journal of Bone and Mineral Research 2011;26(Suppl 1):S223.
RISAROS 2009 {published data only}
-
- EUCTR2006-006943-29. Randomized, double-blind, placebo-controlled trial evaluating the effectiveness of oral risedronate 35 mg per week in the prevention of bone loss in women with breast cancer treated with aromatase inhibitors - Risaros [Essai randomise, en double aveugle contre placebo evaluant l'efficacite du risedronate oral 35 mg par semaine dans la prevention de la perte osseuse chez la femme atteinte d'un cancer du sein traite par inhibiteurs de l'aromatase - Risaros]. https://www.clinicaltrialsregister.eu/ctr-search/search?query=eudract_nu... (date received 2009).
-
- NCT00859703. Study to assess efficacy of risedronate in preventing bone loss in postmenopausal women treated for breast cancer. https://clinicaltrials.gov/ct2/show/NCT00859703 (date received 11 March 2009).
Xu 2010 {published data only}
-
- Xu L, Hao X, Zhang M, Zhang J. Clinical trial on the efficacy of zoledronic acid in preventing bone loss induced by aromatase inhibitor in breast cancer. Chinese Journal of Clinical Oncology 2010;37:392-4.
Yonehara 2007 {published data only}
-
- Yonehara Y, Iwamoto I, Kosha S, Rai Y, Sagara Y, Douchi T. Aromatase inhibitor-induced bone mineral loss and its prevention by bisphosphonate administration in postmenopausal breast cancer patients. Journal of Obstetrics and Gynaecology Research 2007;33:696-9. - PubMed
References to ongoing studies
D‐BIOMARK {published data only}
-
- EUCTR2016-002678-11-ES. An open label biomarker pilot study of the antitumoral acrivity of denosumab in the pre-operative setting of early breast cancer. https://www.clinicaltrialsregister.eu/ctr-search/trial/2016-002678-11/ES (date received 2018).
-
- Ippoliti G, Vethencourt A, Eva T, Feu Llaurado A, Taco C, Guerra E, et al. Denosumab as inmunomodulator in early breast cancer: preliminary results of a randomized window of opportunity clinical trial DBIOMARK (NCT03691311). Virchows Archiv 2021;479(Suppl 1):68‐9.
-
- NCT03691311. Biomarker study of the antitumoral activity of denosumab in the pre-operative setting of early breast cancer. Https://clinicaltrials.gov/show/NCT03691311 (date received 1 October 2018).
-
- Vethencourt A, Trinidad EM, Petit A, Soler-Monso MT, Aleza CG, Urruticochea A, et al. First results of the randomized window of opportunity clinical trial D-Biomark: immunomodulatory effect of denosumab in early breast cancer. Cancer Research 2022;82(4 Suppl):P2-08-10.
-
- Vethencourt AC, Trinidad EM, Gomez Aleza C, Pernas Simon S, Petit A, Soler T, et al. 14P Immunomodulatory effect of denosumab in early breast cancer: preliminary results of a randomized window-opportunity clinical trial D-Biomark. Annals of Oncology 2021;32:26.
ENDEAVOR {published data only}
-
- JprnUminR000024427. A multicenter, cooperative, randomized, comparative study regarding the efficacy of denosumab for bone loss related to postoperative endocrine therapy in postmenopausal patients with hormone-sensitive breast cancer. https://center6.umin.ac.jp/cgi-open-bin/ctr_e/ctr_view.cgi?recptno=R0000... (date received 2016).
-
- NCT03324932. Efficacy of denosumab on normal BMD in women receiving adjuvant aromatase inhibitors for early breast cancer. https://clinicaltrials.gov/ct2/show/NCT03324932 (date received 30 October 2017).
Additional references
Aft 2012
Balduzzi 2019
Bekker 2004
-
- Bekker PJ, Holloway DL, Rasmussen AS, Murphy R, Martin SW, Leese PT, et al. A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. Journal of Bone and Mineral Research 2004;19(7):1059-66. - PubMed
Bray 2018
-
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer Journal for Clinicians 2018;68(6):394-424. - PubMed
Chaimani 2012
-
- Chaimani A, Salanti G. Using network meta-analysis to evaluate the existence of small-study effects in a network of interventions. Research Synthesis Methods 2012;3:161-76. - PubMed
Covidence [Computer program]
-
- Covidence. Version accessed 03/10/2019. Melbourne, Australia: Veritas Health Innovation, 2022. Available at www.covidence.org.
Deeks 2021
-
- Deeks JJ, Higgins JPT, Altman DG (editors). Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins JP, ThomasJ, Chandler J, Cumpston M, Li T, Page MJ, et al (editors). Cochrane Handbook for Systematic Reviews of Interventions version 6.2 (updated February 2021). Cochrane, 2021. Availablefrom training.cochrane.org/handbook/archive/v6.2..
Dhesy‐Thind 2017
-
- Dhesy-Thind S, Fletcher GG, Blanchette PS, Clemons MJ, Dillmon MS, Frank ES, et al. Use of adjuvant bisphosphonates and other bone-modifying agents in breast cancer: a Cancer Care Ontario and American Society of Clinical Oncology clinical practice guideline. Journal of Clinical Oncology 2017;35(18):2062-81. - PubMed
Dias 2010
-
- Dias S, Welten NJ, Caldwell DM, Ades AE. Checking consistency in mixed treatment comparison meta-analysis. Statistics in Medicine 2010;29(7-8):932-44. - PubMed
EBCTCG 2015
-
- Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Adjuvant bisphosphonate treatment in early breast cancer: meta-analyses of individual patient data from randomised trials. Lancet 2015;386(10001):1353-61. - PubMed
Edwards 2013
Egger 1997
Furukawa 2006
-
- Furukawa TA, Barbui C, Cipriani A, Brambilla P, Watanabe N. Imputing missing standard deviations in meta-analyses can provide accurate results. Journal of Clinical Epidemiology 2006;59(1):7-10. - PubMed
Gnant 2009
-
- Gnant M, Mlineritsch B, Schippinger W, Luschin-Ebengreuth G, Postlberger S, Menzel C, et al. Endocrine therapy plus zoledronic acid in premenopausal breast cancer. New England Journal of Medicine 2009;360(7):679-91. - PubMed
Gnant 2011
-
- Gnant M, Mlineritsch B, Stoeger H, Luschin-Ebengreuth G, Heck D, Menzel C, et al. Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 62-month follow-up from the ABCSG-12 randomised trial. Lancet Oncology 2011;12(7):631-41. - PubMed
Gnant 2015
-
- Gnant M, Pfeiler G, Dubsky PC, Hubalek M, Greil R, Jakesz R, et al. Adjuvant denosumab in breast cancer (ABCSG-18): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 2015;386(9992):433-43. - PubMed
Greep 2003
-
- Greep NC, Giuliano AE, Hansen NM, Taketani T, Wang HJ, Singer FR. The effects of adjuvant chemotherapy on bone density in postmenopausal women with early breast cancer. American Journal of Medicine 2003;114(8):653-9. - PubMed
Hadji 2009
-
- Hadji P, Ziller M, Maskow C, Albert U, Kalder M. The influence of chemotherapy on bone mineral density, quantitative ultrasonometry and bone turnover in pre-menopausal women with breast cancer. European Journal of Cancer 2009;45(18):3205-12. - PubMed
Hadji 2011
-
- Hadji P, Asmar L, Van Nes JG, Menschik T, Hasenburg A, Kuck J, et al. The effect of exemestane and tamoxifen on bone health within the Tamoxifen Exemestane Adjuvant Multinational (TEAM) trial: a meta-analysis of the US, German, Netherlands, and Belgium sub-studies. Journal of Cancer Research and Clinical Oncology 2011;137(6):1015-25. - PMC - PubMed
Hadji 2014
-
- Hadji P, Kauka A, Ziller M, Birkholz K, Baier M, Muth M, et al. Effects of zoledronic acid on bone mineral density in premenopausal women receiving neoadjuvant or adjuvant therapies for HR+ breast cancer: the ProBONE II study. Osteoporosis International 2014;25(4):1369-78. - PubMed
Hadji 2017
-
- Hadji P, Aapro MS, Body JJ, Gnant M, Brandi ML, Reginster JY, et al. Management of Aromatase Inhibitor-associated Bone Loss (AIBL) in postmenopausal women with hormone sensitive breast cancer: joint position statement of the IOF, CABS, ECTS, IEG, ESCEO IMS, and SIOG. Journal of Bone Oncology 2017;7:1-12. - PMC - PubMed
Higgins 2011
-
- Higgins JP, Altman DG, Sterne JAC. Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Green S (eds). Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 (updated March 2011). The Cochrane Callaboration, 2011. Available from www.training.cochrane.org/handbook/archive/v5.1.
Higgins 2021
-
- Higgins JPT, Eldridge S, Li T (editors). Chapter 23: Including variants on randomised trials. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archivev6.2.
Hsu 1999
-
- Hsu H, Lacey DL, Dunstan CR, Solovyev I, Colombero A, Timms E, et al. Tumor necrosis factor receptor family member RANK mediates osteoclast differentiation and activation induced by osteoprotegerin ligand. Proceedings of the National Academy of Sciences of the United States of America 1999;96(7):3540-5. - PMC - PubMed
Kalder 2014
-
- Kalder M, Hans D, Kyvernitakis I, Lamy O, Bauer M, Hadji P. Effects of exemestane and tamoxifen treatment on bone texture analysis assessed by TBS in comparison with bone mineral density assessed by DXA in women with breast cancer. Journal of Clinical Densitometry 2014;17(1):66-71. - PubMed
Kanis 2003
-
- Kanis JA, Oden A, Johnell O, De Laet C, Jonsson B, Oglesby AK. The components of excess mortality after hip fracture. Bone 2003;32(5):468-73. - PubMed
Kanis 2008
Khan 2011
Li 2021
-
- Li T, Higgins JPT, Deeks JJ (editors). Chapter 5: Collecting data. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al (editors). Cochrane Handbook for Systematic Reviews of Interventions version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archivev6.2.
Marshall 2018
McDonald 2017
-
- McDonald S, Noel-Storr AH, Thomas J. Harnessing the efficiencies of machine learning and Cochrane Crowd to identify randomised trials for individual Cochrane Reviews. In: Global Evidence Summit, Cape Town, South Africa; 2017. (accessed prior to 31 May, 2024).
Moher 2009
National Guideline Alliance 2018
-
- National Institute for Health and Care Excellence. Early and locally advanced breast cancer: diagnosis and management (NG101). https://www.nice.org.uk/guidance/ng101/resources/early-and-locally-advan... 2018 (updated 2024) (accessed prior to 31 May 2024). - PubMed
NCI 2015
-
- National Cancer Institute. Cancer staging. https://www.cancer.gov/about-cancer/diagnosis-staging/staging (accessed prior to 31 May 2024).
netmeta 2023
-
- Balduzzi S, Rücker G, Nikolakopoulou A, Papakonstantinou T, Salanti G, Efthimiou O, et al. netmeta: An R Package for Network Meta-Analysis Using Frequentist Methods. R package version 2.0-1.. Journal of Statistical Software 2023;106(2):1-40. [DOI: 10.18637/jss.v106.i02] - DOI
Nicolopoulos 2023
-
- Nicolopoulos K, Moshi MR, Stringer D, Ma N, Jenal M, Vreugdenburg T. The clinical effectiveness of denosumab (Prolia R) in patients with hormone-sensitive cancer receiving endocrine therapy, compared to bisphosphonates, selective estrogen receptor modulators (SERM), and placebo: a systematic review and network meta-analysis. Archives of Osteoporosis 2023;18(1):18. - PubMed
Noel‐Storr 2018
-
- Noel-Storr AH, Project Transform team. Cochrane Crowd: new ways of working together to produce health evidence. In: Evidence Live 2018, Oxford, UK. 2018.
O'Carrigan 2017
Parmar 1998
-
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Statistics in Medicine 1998;17(24):2815-34. - PubMed
Puhan 2014
-
- Puhan MA, Schunemann HJ, Murad MH, Li T, Brignardello-Petersen R, Singh JA, et al. A GRADE working group approach for rating the quality of treatment effect estimates from network meta-analysis. British Medical Journal (Clinical research ed.) 2014;349:g5630. - PubMed
R 2021 [Computer program]
-
- R: A language and environment for statistical computing. R Core Team. Vienna, Austria: R Foundation for Statistical Computing, 2021. Available at https://www.R-project.org/.
Rabaglio 2009
RevMan Web 2023 [Computer program]
-
- Review Manager Web (RevMan Web). Version 5.6.0. The Cochrane Collaboration, 2023. Available at revman.cochrane.org.
Reyes 2016
-
- Reyes C, Hitz M, Prieto-Alhambra D, Abrahamsen B. Risks and benefits of bisphosphonate therapies. Journal of Cellular Biochemistry 2016;117(1):20-8. - PubMed
Rodan 1998
Rücker 2012
-
- Rücker G. Network meta-analysis, electrical networks and graph theory. Research Synthesis Methods 2012;3(4):312-24. - PubMed
Rücker 2014
-
- Rücker G, Schwarzer G. Reduce dimension or reduce weights? Comparing two approaches to multi-arm studies in network meta-analysis. Statistics in Medicine 2014;33(25):4353-69. - PubMed
Rücker 2015
Schuenemann 2021
-
- Schunemann HJ, Oxman AD, Vist GE, Higgins JPT, Deeks JJ, Glaziou P, et al. Chapter 14: Completing ‘Summary of findings’ tables and grading the certainty of the evidence. In: Cochrane Handbook for Systematic Reviews of Interventions Version 6.2. Cochrane, 2021. Available from www.training.cochrane.org/handbook/archive/v6.2.
Schwarzer 2015
-
- Schwarzer G, Carpenter JR, Rücker G. Chapter 8: Network meta-analysis. In: Meta-Analysis with R. Switzerland: Springer International Publishing, 2015.
Sterne 2011
-
- Sterne JAC, Egger M, Moher D. Chapter 10: Addressing reporting biases. In: Higgins JPT, Green S, editor(s). Cochrane Handbook of Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.training.cochrane.org/handbook/archive/v5.1.
Tesfamariam 2019
-
- Tesfamariam Y, Jakob T, Wöckel A, Adams A, Weigl A, Monsef I, et al. Adjuvant bisphosphonates or RANK-ligand inhibitors for patients with breast cancer and bone metastases: a systematic review and network meta-analysis. Critical Reviews in Oncology/Hematology 2019;137:1-8. - PubMed
Thomas 2017
-
- Thomas J, Noel-Storr A, Marshall I, Wallace B, McDonald S, Mavergames C, et al. Living systematic reviews: 2. Combining human and machine effort. Journal of Clinical Epidemiology 2017;91:31-7. - PubMed
Tierney 2007
Valachis 2011
-
- Valachis A, Nearchou A, Polyzos NP, Mauri D, Lind PA. Adjuvant therapy with zoledronic acid in primary breast cancer: a systematic review and meta-analysis. European Journal of Cancer 2011;47(Suppl. 1):S377.
Vidal 2012
Yasuda 1998
-
- Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S, et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proceedings of the National Academy of Sciences of the United States of America 1998;95(7):3597-602. - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

