Vilaprisan for the treatment of symptomatic endometriosis: results from a terminated phase 2b randomized controlled trial
- PMID: 38983729
- PMCID: PMC11228778
- DOI: 10.1016/j.xfre.2024.03.002
Vilaprisan for the treatment of symptomatic endometriosis: results from a terminated phase 2b randomized controlled trial
Abstract
Objective: To evaluate the efficacy and safety of 2 doses of vilaprisan vs. placebo in participants with symptomatic endometriosis.
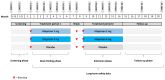
Design: Multicenter, randomized, double-blind, placebo-controlled, parallel-group phase 2b trial (NCT03573336). The initially planned sample size was 315 patients. Recruitment was paused to assess long-term toxicity findings in rodents; although the findings were assessed as likely to be of limited clinical relevance in humans, the study was closed by the sponsor. During the pause, enrolled patients completed 3 or 6 months of treatment per their assigned regimen.
Setting: University hospitals, a regional hospital, and a private clinic.
Patients: Premenopausal adults with confirmed endometriosis and moderate-to-severe pelvic pain (≥4/10 on a numerical rating scale) were enrolled. Inclusion required protocol adherence, including ≥24 diary entries, and an average pain score of ≥3.5.
Intervention: Participants were randomly assigned 1:1:1 to receive vilaprisan (2 mg), vilaprisan (4 mg), or placebo.
Main outcome measures: The primary outcome was a change in the 7-day mean "worst pain" (per the endometriosis symptom diary item 1) from baseline to month 3. All analyses were descriptive only.
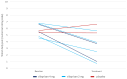
Results: Eight participants were randomly assigned to treatment before the study pause: 6 received vilaprisan (4 mg, n = 4 and 2 mg, n = 2), and 2 received placebo. The 6 vilaprisan recipients experienced an improvement in endometriosis-associated pelvic pain, whereas the 2 placebo recipients experienced no change or increased pain; all 8 participants had decreased use of pain medication. Bleeding intensity decreased from baseline in the vilaprisan group.
Conclusion: The study findings suggest that vilaprisan may improve outcomes in patients with endometriosis. Further studies in larger populations would be needed to accurately assess treatment effects.
Clinical trial registration number: NCT03573336.
Keywords: Vilaprisan; dysmenorrhea; endometriosis; pelvic pain; selective progesterone receptor modulator.
© 2024 The Authors.
Conflict of interest statement
H.S.T. reports having served as an investigator for previous Bayer-sponsored studies and also reports funding from Abbvie and consulting fees from Organon outside the submitted. L.D. reports having served as a full-time employee of Bayer at the time of the study and also reports funding from Bayer for the submitted work and funding and stock options from Bayer outside the submitted work. J.H. reports having served as an investigator for previous Bayer-sponsored studies and also reports funding from Bayer for the submitted work. P.O. reports having served as an investigator for previous Bayer-sponsored studies and also reports to the advisory board for Ryeqo outside of the submitted work. K.T. reports having served as an investigator for previous Bayer-sponsored studies and reports stock options to the Austrian Society of OBGYN outside the submitted work. R.W. reports having served as an investigator for previous Bayer-sponsored studies and also reports consulting fees from Gedeon Richter; honoraria from Gedeon Richter and Intuitive; patents for noninvasive diagnosis of endometriosis; and stock options from Novartis outside the submitted work. T.F. reports having served as a full-time employee of Bayer at the time of the study. E.G.W. reports being a full-time employee of Bayer. X.R. reports being a full-time employee of Bayer. C.S. reports being a full-time employee of Bayer. C.S. also reports funding from Bayer AG Berlin, Germany, for the submitted work; patents on use issued by vilaprisan; and stock options issued by Bayer AG outside the submitted work.
Figures


References
-
- Taylor H.S., Kotlyar A.M., Flores V.A. Endometriosis is a chronic systemic disease: clinical challenges and novel innovations. Lancet. 2021;397:839–852. - PubMed
-
- Hans Evers J.L. Is adolescent endometriosis a progressive disease that needs to be diagnosed and treated? Hum Reprod. 2013;28:2023. - PubMed
-
- Netter A., d'Avout-Fourdinier P., Agostini A., Chanavaz-Lacheray I., Lampika M., Farella M., et al. Progression of deep infiltrating rectosigmoid endometriotic nodules. Hum Reprod. 2019;34:2144–2152. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials

