Myeloid‑derived suppressor cells as targets of emerging therapies and nanotherapies (Review)
- PMID: 38983795
- PMCID: PMC11228699
- DOI: 10.3892/mi.2024.170
Myeloid‑derived suppressor cells as targets of emerging therapies and nanotherapies (Review)
Abstract
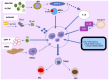
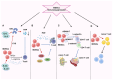
Breast cancer (BC) is the leading cause of cancer-related mortality among women worldwide. Immunotherapies are a promising approach in cancer treatment, particularly for aggressive forms of BC with high mortality rates. However, the current eligibility for immunotherapy remains limited to a limited fraction of patients with BC. Myeloid-derived suppressor cells (MDSCs), originating from myeloid cells, are known for their dual role in immunosuppression and tumor promotion, significantly affecting patient outcomes by fostering the formation of premetastatic niches. Consequently, targeting MDSCs has emerged as a promising avenue for further exploration in therapeutic interventions. Leveraging nanotechnology-based drug delivery systems, which excel in accumulating drugs within tumors via passive or active targeting mechanisms, are a promising strategy for the use of MDSCs in the treatment of BC. The present review discusses the immunosuppressive functions of MDSCs, their role in BC, and the diverse strategies for targeting them in cancer therapy. Additionally, the present review discusses future advancements in BC treatments focusing on MDSCs. Furthermore, it elucidates the mechanisms underlying MDSC activation, recruitment and differentiation in BC progression, highlighting the clinical characteristics that render MDSCs suitable candidates for the therapy and targeted nanotherapy of BC.
Keywords: breast cancer; immunosuppression; immunotherapy; myeloid-derived suppressor cells; nanotherapy; tumor microenvironment.
Copyright: © 2024 Kumar et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Langeh U, Kumar V, Ahuja P, Singh C, Singh A. An update on breast cancer chemotherapy-associated toxicity and their management approaches. Health Sci Re. 2023;9(100119)
Publication types
LinkOut - more resources
Full Text Sources
