Aspirin Dosing for Secondary Prevention of Atherosclerotic Cardiovascular Disease in Male and Female Patients: A Secondary Analysis of the ADAPTABLE Randomized Clinical Trial
- PMID: 38985488
- PMCID: PMC11238071
- DOI: 10.1001/jamacardio.2024.1712
Aspirin Dosing for Secondary Prevention of Atherosclerotic Cardiovascular Disease in Male and Female Patients: A Secondary Analysis of the ADAPTABLE Randomized Clinical Trial
Abstract
Importance: Atherosclerotic cardiovascular disease (ASCVD) remains the leading cause of morbidity and mortality in the US. Although aspirin is recommended for secondary prevention of ASCVD, there was no difference in safety and effectiveness of aspirin dosed daily at 81 mg or 325 mg in the ADAPTABLE (Aspirin Dosing: A Patient-Centric Trial Assessing Benefits and Long-Term Effectiveness) randomized clinical trial. However, it is unknown whether differences by sex exist in the safety and effectiveness of the different aspirin doses.
Objective: To evaluate sex-specific differences in the safety and effectiveness of 2 aspirin doses in the ADAPTAPLE trial.
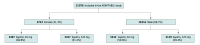
Design, setting, and participants: The ADAPTABLE study was an open-label, pragmatic, randomized clinical trial that randomly assigned participants with chronic, stable ASCVD to 81 mg vs 325 mg of aspirin daily. Using Cox proportional-hazard models, male and female participants were compared for outcomes. In addition, it was assessed whether sex was an effect modifier in the association between aspirin dose and outcomes. The ADAPTABLE trial was conducted at 40 medical centers and 1 health plan. Eligible patients were 18 years and older and had established ASCVD. Study data were analyzed from December 2021 to March 2024.
Interventions: Patients received 81 mg or 325 mg of aspirin daily for the secondary prevention of ASCVD.
Main outcomes and measures: The primary effectiveness outcomes included all-cause death and hospitalization for myocardial infarction (MI) or stroke. The primary safety outcome was hospitalization for major bleeding requiring transfusion.
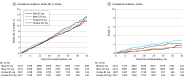
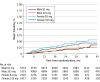
Results: A total of 15 076 patients (median [IQR] age, 67.6 [60.7-73.6] years; 10 352 male [68.7%]) were followed up for a median (IQR) of 26.2 (19.0-34.9) months. Overall, 4724 (31.3%) were female, and 2307 of the female participants (48.8%) received aspirin 81 mg. Compared with males, female participants were younger (median [IQR] age, 66.3 [59.4-72.6] years vs 68.2 (61.4-73.9) years, less likely to self-report White race (3426 [72.5%] vs 8564 [82.7%]), more likely to smoke (564 [12.9%] vs 818 [8.4%]), and more likely to have a history of peripheral arterial disease (1179 [25.7%] vs 2314 [23.0%]). The primary effectiveness outcome of all-cause death and hospitalization for MI or stroke occurred in 379 female participants (8.1%) and 780 male participants (7.1%). There was no significant interaction by sex for the primary effectiveness end point between the 2 aspirin doses (female adjusted hazard ratio [aHR], 1.01; 95% CI, 0.82-1.26 and male aHR, 1.06; 95% CI, 0.91-1.23; P interaction term for sex = .74). During the trial, female participants had fewer revascularization procedures (237 [5.0%] vs 680 [6.6%]; aHR, 0.79; 95% CI, 0.68-0.92; P = .002) but had a higher risk of hospitalization for stroke (aHR, 1.72; 95% CI, 1.27-2.33; P < .001). Among female participants, there was a slightly higher rate of bleeding in the 81-mg aspirin cohort compared with the 325-mg cohort (20 [0.83%] vs 13 [0.52%]; aHR, 2.21; 95% CI, 1.04-4.70; P interaction term for sex = .07). There were no significant differences between female and male participants regarding aspirin dose adherence.
Conclusions and relevance: In this secondary analysis of the ADAPTABLE trial, there were no significant sex-specific differences in the effectiveness and safety of 2 aspirin doses for secondary prevention of ASCVD events.
Trial registration: ClinicalTrials.gov Identifier: NCT02697916.
Conflict of interest statement
Figures
References
-
- Virani SS, Alonso A, Benjamin EJ, et al. ; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee . Heart disease and stroke statistics—2020 update: a report from the American Heart Association. Circulation. 2020;141(9):e139-e596. doi: 10.1161/CIR.0000000000000757 - DOI - PubMed
-
- Gaziano JM, Brotons C, Coppolecchia R, et al. ; ARRIVE Executive Committee . Use of aspirin to reduce risk of initial vascular events in patients at moderate risk of cardiovascular disease (ARRIVE): a randomized, double-blind, placebo-controlled trial. Lancet. 2018;392(10152):1036-1046. doi: 10.1016/S0140-6736(18)31924-X - DOI - PMC - PubMed
-
- Baigent C, Blackwell L, Collins R, et al. ; Antithrombotic Trialists’ (ATT) Collaboration . Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomized trials. Lancet. 2009;373(9678):1849-1860. doi: 10.1016/S0140-6736(09)60503-1 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

