Differential components of bradykinesia in Parkinson's disease revealed by deep brain stimulation
- PMID: 38985938
- PMCID: PMC11427043
- DOI: 10.1152/jn.00320.2022
Differential components of bradykinesia in Parkinson's disease revealed by deep brain stimulation
Abstract
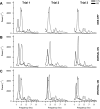
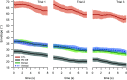
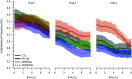
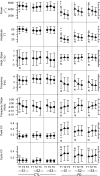
Bradykinesia is a term describing several manifestations of movement disruption caused by Parkinson's disease (PD), including movement slowing, amplitude reduction, and gradual decrease of speed and amplitude over multiple repetitions of the same movement. Deep brain stimulation (DBS) of the subthalamic nucleus (STN) improves bradykinesia in patients with PD. We examined the effect of DBS on specific components of bradykinesia when applied at two locations within the STN, using signal processing techniques to identify the time course of amplitude and frequency of repeated hand pronation-supination movements performed by participants with and without PD. Stimulation at either location increased movement amplitude, increased frequency, and decreased variability, though not to the range observed in the control group. Amplitude and frequency showed decrement within trials, which was similar in PD and control groups and did not change with DBS. Decrement across trials, by contrast, differed between PD and control groups, and was reduced by stimulation. We conclude that DBS improves specific aspects of movement that are disrupted by PD, whereas it does not affect short-term decrement that could reflect muscular fatigue.NEW & NOTEWORTHY In this study, we examined different components of bradykinesia in patients with Parkinson's disease (PD). We identified different components through signal processing techniques and their response to deep brain stimulation (DBS). We found that some components of bradykinesia respond to stimulation, whereas others do not. This knowledge advances our understanding of brain mechanisms that control movement speed and amplitude.
Keywords: DBS; kinematics; motor control; movement; wearable sensors.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures



References
Publication types
MeSH terms
Grants and funding
- R01 NS039913/NS/NINDS NIH HHS/United States
- NS041509/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- NS075321/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- R01 NS107281/NS/NINDS NIH HHS/United States
- R01 NS124789/NS/NINDS NIH HHS/United States
- R01 NS075321/NS/NINDS NIH HHS/United States
- NS075527/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- American Parkinson Disease Association (APDA)
- R01 NS058714/NS/NINDS NIH HHS/United States
- RF1 NS075321/NS/NINDS NIH HHS/United States
- NS058714/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- NS039913/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- K23 NS121630/NS/NINDS NIH HHS/United States
- NS107281/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- NS050425/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- R01 NS103957/NS/NINDS NIH HHS/United States
- R01 NS041509/NS/NINDS NIH HHS/United States
- NS103957/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- NS124789/HHS | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- R01 NS075527/NS/NINDS NIH HHS/United States
- R01 NS050425/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical

