α-Halocarbonyls as a Valuable Functionalized Tertiary Alkyl Source
- PMID: 38989712
- PMCID: PMC12056945
- DOI: 10.1002/open.202400108
α-Halocarbonyls as a Valuable Functionalized Tertiary Alkyl Source
Abstract
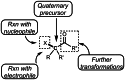
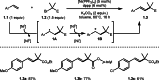
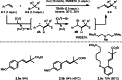
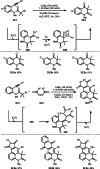
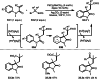
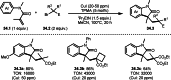
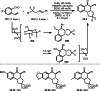
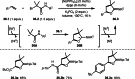
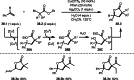
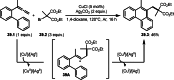
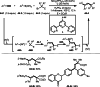
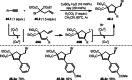
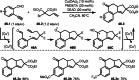
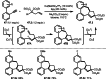
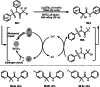
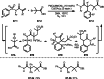
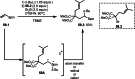
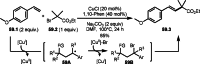
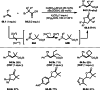
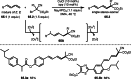
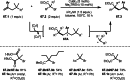
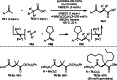
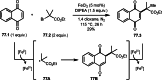
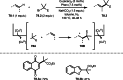
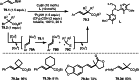
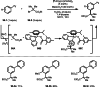
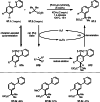
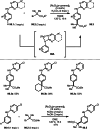
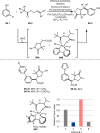
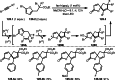
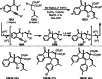
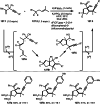
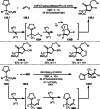
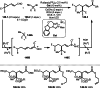
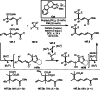
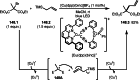
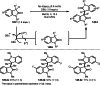
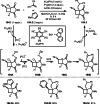
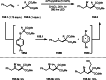
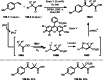
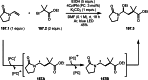
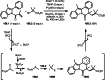
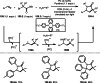
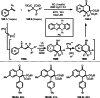
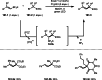
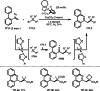
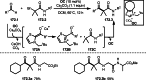
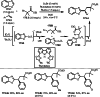
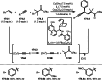
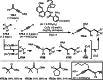
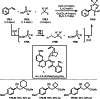
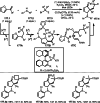
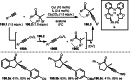
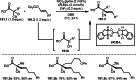
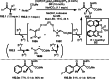
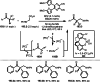
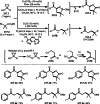
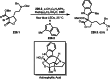
This review introduces the synthetic organic chemical value of α-bromocarbonyl compounds with tertiary carbons. This α-bromocarbonyl compound with a tertiary carbon has been used primarily only as a radical initiator in atom transfer radical polymerization (ATRP) reactions. However, with the recent development of photo-radical reactions (around 2010), research on the use of α-bromocarbonyl compounds as tertiary alkyl radical precursors became popular (around 2012). As more examples were reported, α-bromocarbonyl compounds were studied not only as radicals but also for their applications in organometallic and ionic reactions. That is, α-bromocarbonyl compounds act as nucleophiles as well as electrophiles. The carbonyl group of α-bromocarbonyl compounds is also attractive because it allows the skeleton to be converted after the reaction, and it is being applied to total synthesis. In our survey until 2022, α-bromocarbonyl compounds can be used to perform a full range of reactions necessary for organic synthesis, including multi-component reactions, cross-coupling, substitution, cyclization, rearrangement, stereospecific reactions, asymmetric reactions. α-Bromocarbonyl compounds have created a new trend in tertiary alkylation, which until then had limited reaction patterns in organic synthesis. This review focuses on how α-bromocarbonyl compounds can be used in synthetic organic chemistry.
Keywords: addition; asymmetric reaction; cross-coupling; tertiary alkyl radical; tertiary alkylation.
© 2024 The Authors. ChemistryOpen published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
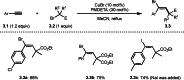
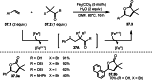
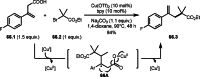


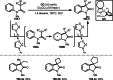
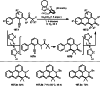
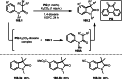
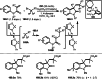
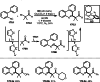



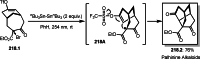
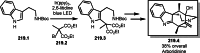

Similar articles
-
Chemistry of Tertiary Carbon Center in the Formation of Congested C-O Ether Bonds.Angew Chem Int Ed Engl. 2021 Feb 19;60(8):4329-4334. doi: 10.1002/anie.202010697. Epub 2020 Dec 22. Angew Chem Int Ed Engl. 2021. PMID: 33350568
-
Controlling alkyne reactivity by means of a copper-catalyzed radical reaction system for the synthesis of functionalized quaternary carbons.Beilstein J Org Chem. 2020 Mar 26;16:502-508. doi: 10.3762/bjoc.16.45. eCollection 2020. Beilstein J Org Chem. 2020. PMID: 32273910 Free PMC article.
-
Synthesizing Complex Quaternary Carbons by the Sequence-Regulated Additions of tert-Alkyl Radicals to Two Different Olefins.Org Lett. 2023 Apr 28;25(16):2840-2845. doi: 10.1021/acs.orglett.3c00812. Epub 2023 Apr 14. Org Lett. 2023. PMID: 37057823
-
Efficient Radical-Mediated Intermolecular α-Alkylation Reactions of Carbonyl Compounds with Nonactivated Alkenes.Chem Asian J. 2024 Jun 17;19(12):e202400319. doi: 10.1002/asia.202400319. Epub 2024 May 14. Chem Asian J. 2024. PMID: 38676345 Review.
-
From Ground-State to Excited-State Activation Modes: Flavin-Dependent "Ene"-Reductases Catalyzed Non-natural Radical Reactions.Acc Chem Res. 2024 May 7;57(9):1446-1457. doi: 10.1021/acs.accounts.4c00129. Epub 2024 Apr 11. Acc Chem Res. 2024. PMID: 38603772 Free PMC article. Review.
Cited by
-
Photoinduced copper-catalysed deracemization of alkyl halides.Nature. 2025 Apr;640(8057):107-113. doi: 10.1038/s41586-025-08784-8. Epub 2025 Apr 2. Nature. 2025. PMID: 40175756
-
A General Platform for Copper-Catalyzed Atom Transfer Radical Addition with Electron-Deficient Olefins.Org Lett. 2025 Jan 10;27(1):241-245. doi: 10.1021/acs.orglett.4c04260. Epub 2024 Dec 18. Org Lett. 2025. PMID: 39694876 Free PMC article.
References
-
- Ling T., Rivas F., Tetrahedron 2016, 72, 6729–6777.
-
- Deno N.C., J. Am. Chem. Soc. 1947, 69, 2233–2234.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

