A transient radial cortical microtubule array primes cell division in Arabidopsis
- PMID: 38990951
- PMCID: PMC11260093
- DOI: 10.1073/pnas.2320470121
A transient radial cortical microtubule array primes cell division in Arabidopsis
Abstract
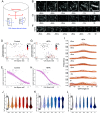
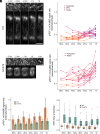
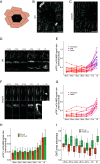
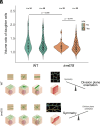
Although the formation of new walls during plant cell division tends to follow maximal tensile stress direction, analyses of individual cells over time reveal a much more variable behavior. The origin of such variability as well as the exact role of interphasic microtubule behavior before cell division have remained mysterious so far. To approach this question, we took advantage of the Arabidopsis stem, where the tensile stress pattern is both highly anisotropic and stable. Although cortical microtubules (CMTs) generally align with maximal tensile stress, we detected a specific time window, ca. 3 h before cell division, where cells form a radial pattern of CMTs. This microtubule array organization preceded preprophase band (PPB) formation, a transient CMT array predicting the position of the future division plane. It was observed under different growth conditions and was not related to cell geometry or polar auxin transport. Interestingly, this cortical radial pattern correlated with the well-documented increase of cytoplasmic microtubule accumulation before cell division. This radial organization was prolonged in cells of the trm678 mutant, where CMTs are unable to form a PPB. Whereas division plane orientation in trm678 is noisier, we found that cell division symmetry was in contrast less variable between daughter cells. We propose that this "radial step" reflects a trade-off in robustness for two essential cell division attributes: symmetry and orientation. This involves a "reset" stage in G2, where an increased cytoplasmic microtubule accumulation transiently disrupts CMT alignment with tissue stress.
Keywords: cell division; cell geometry; mechanical stress; microtubules; robustness.
Conflict of interest statement
Competing interests statement:The authors declare no competing interest.
Figures

Similar articles
-
A role for katanin in plant cell division: microtubule organization in dividing root cells of fra2 and lue1Arabidopsis thaliana mutants.Cytoskeleton (Hoboken). 2011 Jul;68(7):401-13. doi: 10.1002/cm.20522. Cytoskeleton (Hoboken). 2011. PMID: 21721142
-
A Plausible Microtubule-Based Mechanism for Cell Division Orientation in Plant Embryogenesis.Curr Biol. 2018 Oct 8;28(19):3031-3043.e2. doi: 10.1016/j.cub.2018.07.025. Epub 2018 Sep 20. Curr Biol. 2018. PMID: 30245102
-
Microtubule Response to Tensile Stress Is Curbed by NEK6 to Buffer Growth Variation in the Arabidopsis Hypocotyl.Curr Biol. 2020 Apr 20;30(8):1491-1503.e2. doi: 10.1016/j.cub.2020.02.024. Epub 2020 Mar 12. Curr Biol. 2020. PMID: 32169210
-
The making of a ring: Assembly and regulation of microtubule-associated proteins during preprophase band formation and division plane set-up.Curr Opin Plant Biol. 2023 Jun;73:102366. doi: 10.1016/j.pbi.2023.102366. Epub 2023 Apr 15. Curr Opin Plant Biol. 2023. PMID: 37068357 Review.
-
Role of nucleation in cortical microtubule array organization: variations on a theme.Plant J. 2013 Jul;75(2):270-7. doi: 10.1111/tpj.12166. Epub 2013 Apr 1. Plant J. 2013. PMID: 23464654 Review.
Cited by
-
Kinesin-12 POK2 polarization is a prerequisite for a fully functional division site and aids cell plate positioning.Nat Commun. 2025 May 14;16(1):4464. doi: 10.1038/s41467-025-58852-w. Nat Commun. 2025. PMID: 40368920 Free PMC article.
References
-
- Alberts B., Molecular Biology of the Cell (W. W. Norton & Company, ed. 7, 2022).
-
- Stewart M. P., et al. , Hydrostatic pressure and the actomyosin cortex drive mitotic cell rounding. Nature 469, 226–230 (2011). - PubMed
-
- Chanet S., Sharan R., Khan Z., Martin A. C., Myosin 2-induced mitotic rounding enables columnar epithelial cells to interpret cortical spindle positioning cues. Curr. Biol. 27, 3350–3358.e3 (2017). - PubMed
-
- Sawidis T., Quader H., Bopp M., Schnepf E., Presence and absence of the preprophase band of microtubules in moss protonemata: A clue to understanding its function? Protoplasma 163, 156–161 (1991).
-
- Traas J., et al. , Normal differentiation patterns in plants lacking microtubular preprophase bands. Nature 375, 676–677 (1995).
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

