Activation of AMPD2 drives metabolic dysregulation and liver disease in mice with hereditary fructose intolerance
- PMID: 38992061
- PMCID: PMC11239681
- DOI: 10.1038/s42003-024-06539-1
Activation of AMPD2 drives metabolic dysregulation and liver disease in mice with hereditary fructose intolerance
Abstract
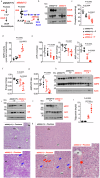
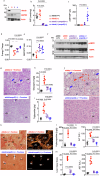
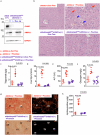
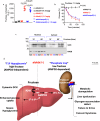
Hereditary fructose intolerance (HFI) is a painful and potentially lethal genetic disease caused by a mutation in aldolase B resulting in accumulation of fructose-1-phosphate (F1P). No cure exists for HFI and treatment is limited to avoid exposure to fructose and sugar. Using aldolase B deficient mice, here we identify a yet unrecognized metabolic event activated in HFI and associated with the progression of the disease. Besides the accumulation of F1P, here we show that the activation of the purine degradation pathway is a common feature in aldolase B deficient mice exposed to fructose. The purine degradation pathway is a metabolic route initiated by adenosine monophosphate deaminase 2 (AMPD2) that regulates overall energy balance. We demonstrate that very low amounts of fructose are sufficient to activate AMPD2 in these mice via a phosphate trap. While blocking AMPD2 do not impact F1P accumulation and the risk of hypoglycemia, its deletion in hepatocytes markedly improves the metabolic dysregulation induced by fructose and corrects fat and glycogen storage while significantly increasing the voluntary tolerance of these mice to fructose. In summary, we provide evidence for a critical pathway activated in HFI that could be targeted to improve the metabolic consequences associated with fructose consumption.
© 2024. The Author(s).
Conflict of interest statement
The authors declare the following competing interests: R.J.J. and M.A.L. have several patents and patent applications related to blocking fructose metabolism in the treatment of metabolic diseases. R.J.J, M.A.L. and D.R.T. are also members of Colorado Research Partners LLC, that is developing inhibitors of fructose metabolism. R.J.J. also has some shares with XORT therapeutics, which is a startup company developing novel xanthine oxidase inhibitors. All other authors declare no competing interests.
Figures


Similar articles
-
Endogenous Fructose Production and Metabolism Drive Metabolic Dysregulation and Liver Disease in Mice with Hereditary Fructose Intolerance.Nutrients. 2023 Oct 16;15(20):4376. doi: 10.3390/nu15204376. Nutrients. 2023. PMID: 37892451 Free PMC article.
-
Aldolase-B knockout in mice phenocopies hereditary fructose intolerance in humans.Mol Genet Metab. 2015 Mar;114(3):445-50. doi: 10.1016/j.ymgme.2015.01.001. Epub 2015 Jan 22. Mol Genet Metab. 2015. PMID: 25637246
-
Ketohexokinase C blockade ameliorates fructose-induced metabolic dysfunction in fructose-sensitive mice.J Clin Invest. 2018 Jun 1;128(6):2226-2238. doi: 10.1172/JCI94427. Epub 2018 Apr 23. J Clin Invest. 2018. PMID: 29533924 Free PMC article.
-
Hereditary fructose intolerance.J Med Genet. 1998 May;35(5):353-65. doi: 10.1136/jmg.35.5.353. J Med Genet. 1998. PMID: 9610797 Free PMC article. Review.
-
The biochemical basis of hereditary fructose intolerance.J Inherit Metab Dis. 2010 Apr;33(2):105-12. doi: 10.1007/s10545-010-9053-2. Epub 2010 Feb 17. J Inherit Metab Dis. 2010. PMID: 20162364 Review.
References
-
- Organization, W. H. Guideline: Sugars intake for adults and children. 1–50 (Genea, 2015). - PubMed
-
- Gitzelmann R, Baerlocher K. Vorteile und Nachteile der Frucosein der Nahrung. Padiat. Fortbildk. Prxis. 1973;37:40–55.
Publication types
MeSH terms
Substances
Grants and funding
- P30 DK048520/DK/NIDDK NIH HHS/United States
- R01 DK121496/DK/NIDDK NIH HHS/United States
- DK108859/U.S. Department of Health & Human Services | NIH | National Institute of Diabetes and Digestive and Kidney Diseases (National Institute of Diabetes & Digestive & Kidney Diseases)
- R01 DK108859/DK/NIDDK NIH HHS/United States
- DK121496/U.S. Department of Health & Human Services | NIH | National Institute of Diabetes and Digestive and Kidney Diseases (National Institute of Diabetes & Digestive & Kidney Diseases)
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

