Establishing delivery route-dependent safety and efficacy of living biodrug mesenchymal stem cells in heart failure patients
- PMID: 38993584
- PMCID: PMC11235206
- DOI: 10.4330/wjc.v16.i6.339
Establishing delivery route-dependent safety and efficacy of living biodrug mesenchymal stem cells in heart failure patients
Abstract
Background: Mesenchymal stem cells (MSCs) as living biopharmaceuticals with unique properties, i.e., stemness, viability, phenotypes, paracrine activity, etc., need to be administered such that they reach the target site, maintaining these properties unchanged and are retained at the injury site to participate in the repair process. Route of delivery (RoD) remains one of the critical determinants of safety and efficacy. This study elucidates the safety and effectiveness of different RoDs of MSC treatment in heart failure (HF) based on phase II randomized clinical trials (RCTs). We hypothesize that the RoD modulates the safety and efficacy of MSC-based therapy and determines the outcome of the intervention.
Aim: To investigate the effect of RoD of MSCs on safety and efficacy in HF patients.
Methods: RCTs were retrieved from six databases. Safety endpoints included mortality and serious adverse events (SAEs), while efficacy outcomes encompassed changes in left ventricular ejection fraction (LVEF), 6-minute walk distance (6MWD), and pro-B-type natriuretic peptide (pro-BNP). Subgroup analyses on RoD were performed for all study endpoints.
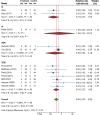
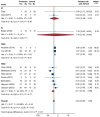
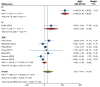
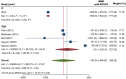
Results: Twelve RCTs were included. Overall, MSC therapy demonstrated a significant decrease in mortality [relative risk (RR): 0.55, 95% confidence interval (95%CI): 0.33-0.92, P = 0.02] compared to control, while SAE outcomes showed no significant difference (RR: 0.84, 95%CI: 0.66-1.05, P = 0.11). RoD subgroup analysis revealed a significant difference in SAE among the transendocardial (TESI) injection subgroup (RR = 0.71, 95%CI: 0.54-0.95, P = 0.04). The pooled weighted mean difference (WMD) demonstrated an overall significant improvement of LVEF by 2.44% (WMD: 2.44%, 95%CI: 0.80-4.29, P value ≤ 0.001), with only intracoronary (IC) subgroup showing significant improvement (WMD: 7.26%, 95%CI: 5.61-8.92, P ≤ 0.001). Furthermore, the IC delivery route significantly improved 6MWD by 115 m (WMD = 114.99 m, 95%CI: 91.48-138.50), respectively. In biochemical efficacy outcomes, only the IC subgroup showed a significant reduction in pro-BNP by -860.64 pg/mL (WMD: -860.64 pg/Ml, 95%CI: -944.02 to -777.26, P = 0.001).
Conclusion: Our study concluded that all delivery methods of MSC-based therapy are safe. Despite the overall benefits in efficacy, the TESI and IC routes provided better outcomes than other methods. Larger-scale trials are warranted before implementing MSC-based therapy in routine clinical practice.
Keywords: Clinical trial; Heart failure; Living biodrug; Mesenchymal stem cells; Meta-analysis; Stem cells; Systematic review.
©The Author(s) 2024. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: The authors declare that they have no conflict of interest to disclose.
Figures


References
-
- Tsao CW, Aday AW, Almarzooq ZI, Alonso A, Beaton AZ, Bittencourt MS, Boehme AK, Buxton AE, Carson AP, Commodore-Mensah Y, Elkind MSV, Evenson KR, Eze-Nliam C, Ferguson JF, Generoso G, Ho JE, Kalani R, Khan SS, Kissela BM, Knutson KL, Levine DA, Lewis TT, Liu J, Loop MS, Ma J, Mussolino ME, Navaneethan SD, Perak AM, Poudel R, Rezk-Hanna M, Roth GA, Schroeder EB, Shah SH, Thacker EL, VanWagner LB, Virani SS, Voecks JH, Wang NY, Yaffe K, Martin SS. Heart Disease and Stroke Statistics-2022 Update: A Report From the American Heart Association. Circulation. 2022;145:e153–e639. - PubMed
-
- Marín-García J. Cell death in the pathogenesis and progression of heart failure. Heart Fail Rev. 2016;21:117–121. - PubMed
-
- Kalou Y, Al-Khani AM, Haider KH. Bone Marrow Mesenchymal Stem Cells for Heart Failure Treatment: A Systematic Review and Meta-Analysis. Heart Lung Circ. 2023;32:870–880. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

