Mild Hyperthermia-Induced Thermogenesis in the Endoplasmic Reticulum Defines Stress Response Mechanisms
- PMID: 38994992
- PMCID: PMC11240596
- DOI: 10.3390/cells13131141
Mild Hyperthermia-Induced Thermogenesis in the Endoplasmic Reticulum Defines Stress Response Mechanisms
Abstract
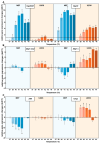
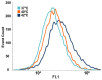
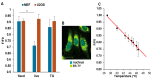
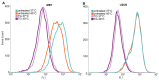
Previous studies reported that a mild, non-protein-denaturing, fever-like temperature increase induced the unfolded protein response (UPR) in mammalian cells. Our dSTORM super-resolution microscopy experiments revealed that the master regulator of the UPR, the IRE1 (inositol-requiring enzyme 1) protein, is clustered as a result of UPR activation in a human osteosarcoma cell line (U2OS) upon mild heat stress. Using ER thermo yellow, a temperature-sensitive fluorescent probe targeted to the endoplasmic reticulum (ER), we detected significant intracellular thermogenesis in mouse embryonic fibroblast (MEF) cells. Temperatures reached at least 8 °C higher than the external environment (40 °C), resulting in exceptionally high ER temperatures similar to those previously described for mitochondria. Mild heat-induced thermogenesis in the ER of MEF cells was likely due to the uncoupling of the Ca2+/ATPase (SERCA) pump. The high ER temperatures initiated a pronounced cytosolic heat-shock response in MEF cells, which was significantly lower in U2OS cells in which both the ER thermogenesis and SERCA pump uncoupling were absent. Our results suggest that depending on intrinsic cellular properties, mild hyperthermia-induced intracellular thermogenesis defines the cellular response mechanism and determines the outcome of hyperthermic stress.
Keywords: RT-qPCR; flow cytometry; fluorescence microscopy; heat-shock response; human osteosarcoma cells; mild heat; mouse embryonic fibroblast cells; organelle temperature; spectrofluorometry; super-resolution microscopy; thermogenesis; unfolded protein response.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Peksel B., Gombos I., Péter M., Vigh L., Tiszlavicz Á., Brameshuber M., Balogh G., Schütz G.J., Horváth I., Vigh L., et al. Mild Heat Induces a Distinct “Eustress” Response in Chinese Hamster Ovary Cells but Does Not Induce Heat Shock Protein Synthesis. Sci. Rep. 2017;7:15643. doi: 10.1038/s41598-017-15821-8. - DOI - PMC - PubMed
-
- Tiszlavicz Á., Gombos I., Péter M., Hegedűs Z., Hunya Á., Dukic B., Nagy I., Peksel B., Balogh G., Horváth I., et al. Distinct Cellular Tools of Mild Hyperthermia-Induced Acquired Stress Tolerance in Chinese Hamster Ovary Cells. Biomedicines. 2022;10:1172. doi: 10.3390/biomedicines10051172. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

