Super T2-FLAIR mismatch sign: a prognostic imaging biomarker for non-enhancing astrocytoma, IDH-mutant
- PMID: 38995493
- PMCID: PMC11341624
- DOI: 10.1007/s11060-024-04758-4
Super T2-FLAIR mismatch sign: a prognostic imaging biomarker for non-enhancing astrocytoma, IDH-mutant
Abstract
Purpose: The T2-FLAIR mismatch sign is a highly specific diagnostic imaging biomarker for astrocytoma, IDH-mutant. However, a definitive prognostic imaging biomarker has yet to be identified. This study investigated imaging prognostic markers, specifically analyzing T2-weighted and FLAIR images of this tumor.
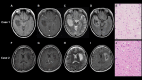
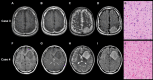
Methods: We retrospectively analyzed 31 cases of non-enhancing astrocytoma, IDH-mutant treated at our institution, and 30 cases from The Cancer Genome Atlas (TCGA)/The Cancer Imaging Archive (TCIA). We defined "super T2-FLAIR mismatch sign" as having a significantly strong low signal comparable to cerebrospinal fluid at non-cystic lesions rather than just a pale FLAIR low-signal tumor lesion as in conventional T2-FLAIR mismatch sign. Cysts were defined as having a round or oval shape and were excluded from the criteria for the super T2-FLAIR mismatch sign. We evaluated the presence or absence of the T2-FLAIR mismatch sign and super T2-FLAIR mismatch sign using preoperative MRI and analyzed the progression-free survival (PFS) and overall survival (OS) by log-rank test.
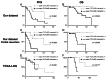
Results: The T2-FLAIR mismatch sign was present in 17 cases (55%) in our institution and 9 cases (30%) within the TCGA-LGG dataset without any correlation with PFS or OS. However, the super T2-FLAIR mismatch sign was detected in 8 cases (26%) at our institution and 13 cases (43%) in the TCGA-LGG dataset. At our institution, patients displaying the super T2-FLAIR mismatch sign showed significantly extended PFS (122.7 vs. 35.9 months, p = 0.0491) and OS (not reached vs. 116.7 months, p = 0.0232). Similarly, in the TCGA-LGG dataset, those with the super T2-FLAIR mismatch sign exhibited notably longer OS (not reached vs. 44.0 months, p = 0.0177).
Conclusion: The super T2-FLAIR mismatch is a promising prognostic imaging biomarker for non-enhancing astrocytoma, IDH-mutant.
Keywords: Astrocytoma; Diffuse glioma; IDH-mutant; Imaging biomarker; Non-enhancing glioma; T2-FLAIR mismatch sign.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
T2-FLAIR mismatch sign, an imaging biomarker for CDKN2A-intact in non-enhancing astrocytoma, IDH-mutant.Neurosurg Rev. 2024 Aug 9;47(1):412. doi: 10.1007/s10143-024-02632-5. Neurosurg Rev. 2024. PMID: 39117984 Free PMC article.
-
Usefulness of synthetic MRI for differentiation of IDH-mutant diffuse gliomas and its comparison with the T2-FLAIR mismatch sign.J Neurooncol. 2024 Nov;170(2):429-436. doi: 10.1007/s11060-024-04794-0. Epub 2024 Aug 12. J Neurooncol. 2024. PMID: 39133381 Free PMC article.
-
The T2-FLAIR mismatch sign as an imaging marker for non-enhancing IDH-mutant, 1p/19q-intact lower-grade glioma: a validation study.Neuro Oncol. 2018 Sep 3;20(10):1393-1399. doi: 10.1093/neuonc/noy048. Neuro Oncol. 2018. PMID: 29590424 Free PMC article.
-
Enhancing glioma care with advanced imaging: T2-FLAIR mismatch as a predictive biomarker in IDH-mutant astrocytoma.Neurosurg Rev. 2024 Aug 30;47(1):512. doi: 10.1007/s10143-024-02748-8. Neurosurg Rev. 2024. PMID: 39212803 Review.
-
The T2-FLAIR-mismatch sign as an imaging biomarker for IDH and 1p/19q status in diffuse low-grade gliomas: a systematic review with a Bayesian approach to evaluation of diagnostic test performance.Neurosurg Focus. 2019 Dec 1;47(6):E13. doi: 10.3171/2019.9.FOCUS19660. Neurosurg Focus. 2019. PMID: 31786548
Cited by
-
Prognostic value of immunohistochemical staining for H3K27me3 and EZH2 in astrocytoma, IDH-mutant.J Neurooncol. 2025 Mar;172(1):185-194. doi: 10.1007/s11060-024-04897-8. Epub 2024 Dec 5. J Neurooncol. 2025. PMID: 39636550 Free PMC article.
-
Diagnosis of Isocitrate Dehydrogenase-Mutant Astrocytoma in the Subcallosal Gyrus Using T2-Fluid-Attenuated Inversion Recovery Mismatch Sign and Quantitative Magnetic Resonance Relaxometry.Cureus. 2025 Jan 5;17(1):e76963. doi: 10.7759/cureus.76963. eCollection 2025 Jan. Cureus. 2025. PMID: 39906460 Free PMC article.
-
Innovation and technology in neurosurgery oncology.J Neurooncol. 2025 May;172(3):499-502. doi: 10.1007/s11060-025-04941-1. Epub 2025 Feb 3. J Neurooncol. 2025. PMID: 39899178 No abstract available.
References
-
- Dubbink HJ, Atmodimedjo PN, Kros JM et al (2016) Molecular classification of anaplastic oligodendroglioma using next-generation sequencing: a report of the prospective randomized EORTC brain Tumor Group 26951 phase III trial. Neuro Oncol 18:388–400. 10.1093/neuonc/nov182 10.1093/neuonc/nov182 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

