CD22-directed CAR T-cell therapy for large B-cell lymphomas progressing after CD19-directed CAR T-cell therapy: a dose-finding phase 1 study
- PMID: 38996463
- PMCID: PMC11329226
- DOI: 10.1016/S0140-6736(24)00746-3
CD22-directed CAR T-cell therapy for large B-cell lymphomas progressing after CD19-directed CAR T-cell therapy: a dose-finding phase 1 study
Abstract
Background: Outcomes are poor for patients with large B-cell lymphoma who relapse after CD19-directed chimeric antigen receptor (CAR) T-cell therapy (CAR19). CD22 is a nearly universally expressed B-cell surface antigen and the efficacy of a CD22-directed CAR T-cell therapy (CAR22) in large B-cell lymphoma is unknown, which was what we aimed to examine in this study.
Methods: In this single centre, open-label, dose-escalation phase 1 trial, we intravenously administered CAR22 at two dose levels (1 million and 3 million CAR22-positive T cells per kg of bodyweight) to adult patients (aged ≥18 years) who relapsed after CAR19 or had CD19-negative large B-cell lymphoma. The primary endpoints were manufacturing feasibility, safety measured by the incidence and severity of adverse events and dose-limiting toxicities, and identification of the maximum tolerated dose (ie, the recommended phase 2 dose). This study is registered with ClinicalTrials.gov (NCT04088890) and is active, but closed for enrolment.
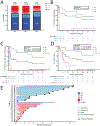
Findings: From Oct 17, 2019, to Oct 19, 2022, a total of 41 patients were assessed for eligibility; however, one patient withdrew. 40 patients underwent leukapheresis and 38 (95%) had CAR T-cell products manufactured successfully. The median age was 65 years (range 25-84), 17 (45%) were women, 32 (84%) had elevated pretreatment lactate dehydrogenase, 11 (29%) had refractory disease to all previous therapies, and patients had received a median of four lines of previous therapy (range 3-8). Of the 38 patients treated, 37 (97%) had relapsed after previous CAR19. The identified maximum tolerated dose was 1 million CAR T cells per kg. Of 29 patients who received the maximum tolerated dose, no patients developed a dose-limiting toxicity or grade 3 or higher cytokine release syndrome, immune effector cell-associated neurotoxicity syndrome, or immune effector cell-associated haemophagocytic lymphohistiocytosis-like syndrome.
Interpretation: This trial identifies CD22 as an immunotherapeutic target in large B-cell lymphoma and demonstrates the durable clinical activity of CAR22 in patients with disease progression after CAR19 therapy. Although these findings are promising, it is essential to recognise that this is a phase 1 dose-finding study. Further investigations are warranted to establish the long-term efficacy and to delineate the patient subgroups that will derive the most benefit from this therapeutic approach.
Funding: National Cancer Institute, National Institutes of Health, Stanford Cancer Institute, Leukemia & Lymphoma Society, Parker Institute for Cancer Immunotherapy, Lymph & Co, and the European Hematology Association.
Copyright © 2024 Elsevier Ltd. All rights reserved, including those for text and data mining, AI training, and similar technologies.
Conflict of interest statement
Declaration of interests ARR received research support from Pharmacyclics and AbbVie; one-time ad hoc scientific advisory board work for Nohla Therapeutics and Kaleido; and expert witness work for the US Department of Justice. CLM is the founder, has equity in, consults for, and is a Director of Cargo Therapeutics and Link Cell Therapies; has equity in Lyell Immunopharma; and receives royalties from the National Institutes of Health for CAR22 consulting for Immatics, Ensoma, Mammoth, Adaptimmune, and Bristol Myers Squibb. DBM consults for Kite Pharma-Gilead, Juno Therapeutics-Celgene, Novartis, Janssen, and Pharmacyclics; and receives research support from Kite Pharma-Gilead, Allogene, Cargo Therapeutics, Pharmacyclics, Miltenyi Biotec, and Adaptive Biotechnologies. JHB consults for Kite Pharma-Gilead; and receives research support from Kite Pharma-Gilead, Genentech-Roche, Regeneron Pharmaceuticals, and Cellular Biomedicine Group. MJF consults for Kite Pharma-Gilead, Adaptative Biotechnologies, and Cargo Therapeutics; receives research support from Kite-Pharma-Gilead, Allogene Therapeutics, Cargo Therapeutics, and Adaptative Biotechnologies; and is on the data safety monitoring board for Fate Therapeutics. LM receives research support from Adaptive Biotechnologies and Servier Laboratories; and consults for Amgen and Pfizer. JYS consults for Kite Pharma-Gilead and ImmPACT Bio. PS receives research support from Kite Pharma-Gilead. SS consults for Janssen. SAF consults for Lonza PerMed, Gradalis, Obsidian, and Samsara BioCapital. MPH is on the Kite Pharma-Gilead advisory board. All other authors declare no competing interests.
Figures


Comment in
-
CD22 CAR T-cell therapy: new hope for patients with large B-cell lymphoma.Lancet. 2024 Jul 27;404(10450):314-315. doi: 10.1016/S0140-6736(24)00815-8. Epub 2024 Jul 9. Lancet. 2024. PMID: 38996464 No abstract available.
References
-
- Locke FL, Miklos DB, Jacobson CA, et al. Axicabtagene Ciloleucel as Second-Line Therapy for Large B-Cell Lymphoma. N Engl J Med 2022;386:640–54. - PubMed
-
- Kamdar M, Solomon SR, Arnason J, et al. Lisocabtagene maraleucel versus standard of care with salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment in patients with relapsed or refractory large B-cell lymphoma (TRANSFORM): results from an interim analysis of an open-label, randomised, phase 3 trial. Lancet (London, England) 2022;399:2294–308. - PubMed
-
- Schuster SJ, Bishop MR, Tam CS, et al. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N Engl J Med 2019;380:45–56. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

