Glycation in the cardiomyocyte
- PMID: 38997172
- PMCID: PMC11578284
- DOI: 10.1016/bs.vh.2024.04.005
Glycation in the cardiomyocyte
Abstract
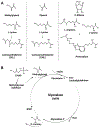
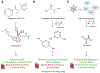
Glycation is a protein post-translational modification that can occur on lysine and arginine residues as a result of a non-enzymatic process known as the Maillard reaction. This modification is irreversible, so the only way it can be removed is by protein degradation and replacement. Small reactive carbonyl species, glyoxal and methylglyoxal, are the primary glycating agents and are elevated in several conditions associated with an increased risk of cardiovascular disease, including diabetes, rheumatoid arthritis, smoking, and aging. Thus, how protein glycation impacts the cardiomyocyte is of particular interest, to both understand how these conditions increase the risk of cardiovascular disease and how glycation might be targeted therapeutically. Glycation can affect the cardiomyocyte through extracellular mechanisms, including RAGE-based signaling, glycation of the extracellular matrix that modifies the mechanical environment, and signaling from the vasculature. Intracellular glycation of the cardiomyocyte can impact calcium handling, protein quality control and cell death pathways, as well as the cytoskeleton, resulting in a blunted contractility. While reducing protein glycation and its impact on the heart has been an active area of drug development, multiple clinical trials have had mixed results and these compounds have not been translated to the clinic-highlighting the challenges of modulating myocyte glycation. Here we will review protein glycation and its effects on the cardiomyocyte, therapeutic attempts to reverse these, and offer insight as to the future of glycation studies and patient treatment.
Keywords: Advanced glycation end-products (AGEs); Cardiomyocyte; Glycation; Glyoxalase; Post-translational modification (PTM); Protein quality control (PQC); Receptors for advanced glycation end-products (RAGE) Note: Also called AGER; Sarcomere.
Copyright © 2024. Published by Elsevier Inc.
Figures
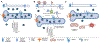


References
-
- Alikhani Z, Alikhani M, Boyd CM, Nagao K, Trackman PC, & Graves DT (2005). Advanced glycation end products enhance expression of pro-apoptotic genes and stimulate fibroblast apoptosis through cytoplasmic and mitochondrial pathways. J Biol Chem, 280(13), 12087–12095. 10.1074/jbc.M406313200 - DOI - PubMed
-
- Arai M, Yuzawa H, Nohara I, Ohnishi T, Obata N, Iwayama Y, Haga S, Toyota T, Ujike H, Arai M, Ichikawa T, Nishida A, Tanaka Y, Furukawa A, Aikawa Y, Kuroda O, Niizato K, Izawa R, Nakamura K, . . . Itokawa M (2010). Enhanced carbonyl stress in a subpopulation of schizophrenia. Arch Gen Psychiatry, 67(6), 589–597. 10.1001/archgenpsychiatry.2010.62 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

