The Simulated Physiological Oocyte Maturation (SPOM) System Enhances Cytoplasmic Maturation and Oocyte Competence in Cattle
- PMID: 38998004
- PMCID: PMC11240716
- DOI: 10.3390/ani14131893
The Simulated Physiological Oocyte Maturation (SPOM) System Enhances Cytoplasmic Maturation and Oocyte Competence in Cattle
Abstract
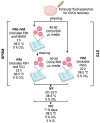
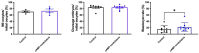
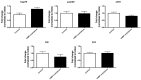
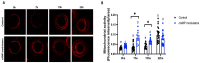
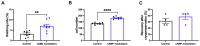
In vitro embryo production is a widely applied technique that allows the expansion of genetics and accelerated breeding programs. However, in cattle, this technique still needs improvement in order to reach quality and pregnancy rates comparable to in vivo-derived embryos. One of the limitations of this technique is related to in vitro maturation, where a heterogeneous population of oocytes is harvested from follicles and cultured in vitro in the presence of gonadotropic hormones to induce maturation. As a result, oocytes with different degrees of competence are obtained, resulting in a decrease in the quality and quantity of embryos obtained. A novel system based on the use of cyclic adenosine monophosphate (cAMP) modulators was developed to enhance bovine oocyte competence, although controversial results were obtained depending on the in vitro embryo production (IVP) system used in each laboratory. Thus, in the present work, we employed a reported cAMP protocol named Simulated Physiological Oocyte Maturation (SPOM) under our IVP system and analysed its effect on cytoplasmic maturation by measuring levels of stress-related genes and evaluating the activity and distribution of mitochondria as a marker for cytoplasmic maturation Moreover, we studied the effect of the cAMP treatment on nuclear maturation, cleavage, and blastocyst formation. Finally, we assessed the embryo quality by determining the hatching rates, total cell number per blastocyst, cryopreservation tolerance, and embryo implantation. We found that maturing oocytes in the presence of cAMP modulators did not affect nuclear maturation, although they changed the dynamic pattern of mitochondrial activity along maturation. Additionally, we found that oocytes subjected to cAMP modulators significantly improved blastocyst formation (15.5% vs. 22.2%, p < 0.05). Blastocysts derived from cAMP-treated oocytes did not improve cryopreservation tolerance but showed an increased hatching rate, a higher total cell number per blastocyst and, when transferred to hormonally synchronised recipients, produced pregnancies. These results reflect that the use of cAMP modulators during IVM results in competent oocytes that, after fertilisation, can develop in more blastocysts with a better quality than standard IVM conditions.
Keywords: bovine; cAMP modulators; cytoplasmic maturation; embryo development; in vitro maturation; mitochondrial activity; oocyte competence.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Simulated physiological oocyte maturation has side effects on bovine oocytes and embryos.J Assist Reprod Genet. 2019 Mar;36(3):413-424. doi: 10.1007/s10815-018-1365-4. Epub 2018 Nov 16. J Assist Reprod Genet. 2019. PMID: 30443692 Free PMC article.
-
Extending prematuration with cAMP modulators enhances the cumulus contribution to oocyte antioxidant defence and oocyte quality via gap junctions.Hum Reprod. 2016 Apr;31(4):810-21. doi: 10.1093/humrep/dew020. Epub 2016 Feb 22. Hum Reprod. 2016. PMID: 26908844
-
A pre-in vitro maturation medium containing cumulus oocyte complex ligand-receptor signaling molecules maintains meiotic arrest, supports the cumulus oocyte complex and improves oocyte developmental competence.Mol Hum Reprod. 2017 Sep 1;23(9):594-606. doi: 10.1093/molehr/gax032. Mol Hum Reprod. 2017. PMID: 28586460
-
The Simulated Physiological Oocyte Maturation (SPOM) system in domestic animals: A systematic review.Theriogenology. 2022 Aug;188:90-99. doi: 10.1016/j.theriogenology.2022.05.023. Epub 2022 Jun 1. Theriogenology. 2022. PMID: 35688043
-
Effects of in vivo prematuration and in vivo final maturation on developmental capacity and quality of pre-implantation embryos.Theriogenology. 2002 Jan 1;57(1):5-20. doi: 10.1016/s0093-691x(01)00655-0. Theriogenology. 2002. PMID: 11775980 Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

