Dendrimer Platforms for Targeted Doxorubicin Delivery-Physicochemical Properties in Context of Biological Responses
- PMID: 39000306
- PMCID: PMC11241532
- DOI: 10.3390/ijms25137201
Dendrimer Platforms for Targeted Doxorubicin Delivery-Physicochemical Properties in Context of Biological Responses
Abstract
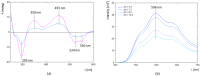
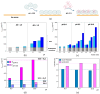
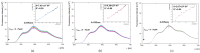
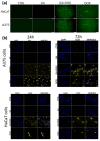
The unique structure of G4.0 PAMAM dendrimers allows a drug to be enclosed in internal spaces or immobilized on the surface. In the conducted research, the conditions for the formation of the active G4.0 PAMAM complex with doxorubicin hydrochloride (DOX) were optimized. The physicochemical properties of the system were monitored using dynamic light scattering (DLS), circular dichroism (CD), and fluorescence spectroscopy. The Quartz Crystal Microbalance with Dissipation Monitoring (QCM-D) method was chosen to determine the preferential conditions for the complex formation. The highest binding efficiency of the drug to the cationic dendrimer was observed under basic conditions when the DOX molecule was deprotonated. The decrease in the zeta potential of the complex confirms that DOX immobilizes through electrostatic interaction with the carrier's surface amine groups. The binding constants were determined from the fluorescence quenching of the DOX molecule in the presence of G4.0 PAMAM. The two-fold way of binding doxorubicin in the structure of dendrimers was visible in the Isothermal calorimetry (ITC) isotherm. Fluorescence spectra and release curves identified the reversible binding of DOX to the nanocarrier. Among the selected cancer cells, the most promising anticancer activity of the G4.0-DOX complex was observed in A375 malignant melanoma cells. Moreover, the preferred intracellular location of the complexes concerning the free drug was found, which is essential from a therapeutic point of view.
Keywords: DDS; PAMAM dendrimers; dendrimer-doxorubicin interactions; doxorubicin; drug delivery systems.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Theoretical and Experimental Analyses of the Interfacial Mechanism of Dendrimer-Doxorubicin Complexes Formation.Mol Pharm. 2024 Nov 4;21(11):5892-5904. doi: 10.1021/acs.molpharmaceut.4c00941. Epub 2024 Oct 22. Mol Pharm. 2024. PMID: 39436101 Free PMC article.
-
Low systemic toxicity nanocarriers fabricated from heparin-mPEG and PAMAM dendrimers for controlled drug release.Mater Sci Eng C Mater Biol Appl. 2018 Jan 1;82:291-298. doi: 10.1016/j.msec.2017.07.051. Epub 2017 Jul 31. Mater Sci Eng C Mater Biol Appl. 2018. PMID: 29025661
-
Selective Cellular Uptake and Druggability Efficacy through Functionalized Chitosan-Conjugated Polyamidoamine (PAMAM) Dendrimers.Sensors (Basel). 2024 Jul 26;24(15):4853. doi: 10.3390/s24154853. Sensors (Basel). 2024. PMID: 39123900 Free PMC article.
-
Review on the targeted conjugation of anticancer drugs doxorubicin and tamoxifen with synthetic polymers for drug delivery.J Biomol Struct Dyn. 2017 Aug;35(11):2497-2508. doi: 10.1080/07391102.2016.1222971. Epub 2016 Sep 6. J Biomol Struct Dyn. 2017. PMID: 27598545 Review.
-
Dendrimer as a promising nanocarrier for the delivery of doxorubicin as an anticancer therapeutics.J Biomater Sci Polym Ed. 2021 Oct;32(14):1882-1909. doi: 10.1080/09205063.2021.1938859. Epub 2021 Jul 20. J Biomater Sci Polym Ed. 2021. PMID: 34078252 Review.
Cited by
-
A Comprehensive Review of Smart Thermosensitive Nanocarriers for Precision Cancer Therapy.Int J Mol Sci. 2025 Jul 29;26(15):7322. doi: 10.3390/ijms26157322. Int J Mol Sci. 2025. PMID: 40806454 Free PMC article. Review.
-
Theoretical and Experimental Analyses of the Interfacial Mechanism of Dendrimer-Doxorubicin Complexes Formation.Mol Pharm. 2024 Nov 4;21(11):5892-5904. doi: 10.1021/acs.molpharmaceut.4c00941. Epub 2024 Oct 22. Mol Pharm. 2024. PMID: 39436101 Free PMC article.
-
Transfection Technologies for Next-Generation Therapies.J Clin Med. 2025 Aug 5;14(15):5515. doi: 10.3390/jcm14155515. J Clin Med. 2025. PMID: 40807133 Free PMC article. Review.
-
Electrospun Fiber Mats with Metronidazole: Design, Evaluation, and Release Kinetics.J Phys Chem B. 2025 May 8;129(18):4535-4546. doi: 10.1021/acs.jpcb.5c00873. Epub 2025 Apr 3. J Phys Chem B. 2025. PMID: 40178488 Free PMC article.
-
A Novel PAMAM G3 Dendrimer-Based Foam with Polyether Polyol and Castor Oil Components as Drug Delivery System into Cancer and Normal Cells.Materials (Basel). 2024 Aug 7;17(16):3905. doi: 10.3390/ma17163905. Materials (Basel). 2024. PMID: 39203083 Free PMC article.
References
-
- Zhu L., Yang S., Qu X., Zhu F., Liang Y., Liang F., Wang Q., Li J., Li Z., Yang Z. Fibril-shaped aggregates of doxorubicin with poly-l-lysine and its derivative. Polym. Chem. 2014;5:5700–5706. doi: 10.1039/C4PY00686K. - DOI
-
- Pyne A., Kundu S., Banerjee P., Sarkar N. Unveiling the Aggregation Behavior of Doxorubicin Hydrochloride in Aqueous Solution of 1-Octyl-3-methylimidazolium Chloride and the Effect of Bile Salt on These Aggregates: A Microscopic Study. Langmuir. 2018;34:3296–3306. doi: 10.1021/acs.langmuir.8b00029. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

