Synthesis, enzyme inhibition assay, and molecular modeling study of novel pyrazolines linked to 4-methylsulfonylphenyl scaffold: antitumor activity and cell cycle analysis
- PMID: 39005246
- PMCID: PMC11240878
- DOI: 10.1039/d4ra03902e
Synthesis, enzyme inhibition assay, and molecular modeling study of novel pyrazolines linked to 4-methylsulfonylphenyl scaffold: antitumor activity and cell cycle analysis
Abstract
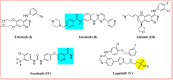
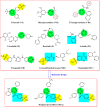
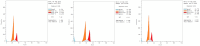
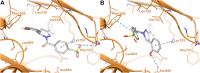
Antitumor activity using 59 cancer cell lines and enzyme inhibitory activity of a newly synthesized pyrazoline-linked 4-methylsulfonylphenyl scaffold (compounds 18a-q) were measured and compared with those of standard drugs. Pyrazolines 18b, 18c, 18f, 18g, 18h, and 18n possessed significant antitumor activity, with a positive cytotoxic effect (PCE) of 22/59, 21/59, 21/59, 48/59, 51/59, and 20/59, respectively. The cancer cell lines HL60, MCF-7, and MDA-MB-231 were used to measure the IC50 values of derivatives 18c, 18g, and 18hvia the MTT assay method, and the results were compared with those of reference drugs. Derivatives 18g and 18h showed potent and broad-spectrum antitumor activities against HL60 (IC50 of 10.43, 8.99 μM, respectively), MCF-7 (IC50 of 11.7 and 12.4 μM, respectively), and MDA-MB-231 (IC50 of 4.07 and 7.18 μM, respectively). Compound 18c exhibited strong antitumor activity against HL60 and MDA-MB-231 cell lines with IC50 values of 8.43 and 12.54 μM, respectively, and moderate antitumor activity against MCF-7 cell lines with an IC50 value of 16.20 μM. Compounds 18c, 18g, and 18h remarkably inhibited VEGFR2 kinase (IC50 = 0.218, 0.168, and 0.135 μM, respectively) compared with the reference drug sorafenib (IC50 = 0.041 μM). Compounds 18g and 18h effectively inhibited HER2 kinase (IC50 = 0.496 and 0.253 μM, respectively) compared with erlotinib (IC50 = 0.085 μM). Compound 18h inhibited EGFR kinase (IC50 = 0.574 μM) with a potency comparable with that of the reference drug erlotinib (IC50 = 0.105 μM). Pyrazolines 18c, 18f, and 18h arrested the S/G2 phase of the cell cycle in HL-60 cells. In addition, derivatives 18c, 18f, and 18h revealed lower Bcl-2 protein expression anti-apoptotic levels and higher Bax, caspase-3, and caspase-9 expression levels. Molecular docking studies of derivative 18h into the binding sites of EGFR, HER2, and VEGFR2 kinases explored the interaction mode of these pyrazoline derivatives and their structural requirements for antitumor activity.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures




References
-
- Avendaño C. and Menendez J. C., Medicinal Chemistry of Anticancer Drugs, Elsevier, 2015
-
- Li Q. Xu W. Anti-Cancer Agents. 2005;5:3–63. - PubMed
-
- Varmus H. Science. 2006;312:1162–1165. - PubMed
-
- Vainshelboim B. Müller J. Lima R. M. Nead K. T. Chester C. Chan K. Kokkinos P. Myers J. Prev. Med. 2017;100:89–94. - PubMed
-
- Bray F. Laversanne M. Weiderpass E. Soerjomataram I. Cancer. 2021;127:3029–3030. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

