Clinical outcomes and predictors of transapical transcatheter mitral valve replacement: the Tendyne Expanded Clinical Study
- PMID: 39007829
- PMCID: PMC11228541
- DOI: 10.4244/EIJ-D-23-00904
Clinical outcomes and predictors of transapical transcatheter mitral valve replacement: the Tendyne Expanded Clinical Study
Abstract
Background: Transcatheter mitral valve replacement (TMVR) is a therapeutic option for patients with severe mitral regurgitation (MR) who are ineligible for conventional surgery. There are limited data on the outcomes of large patient cohorts treated with TMVR.
Aims: This study aimed to investigate the outcomes and predictors of mortality for patients treated with transapical TMVR.
Methods: This analysis represents the clinical experience of all patients enrolled in the Tendyne Expanded Clinical Study. Patients with symptomatic MR underwent transapical TMVR with the Tendyne system between November 2014 and June 2020. Outcomes and adverse events up to 2 years, as well as predictors of short-term mortality, were assessed.
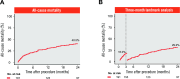
Results: A total of 191 patients were treated (74.1±8.0 years, 62.8% male, Society of Thoracic Surgeons Predicted Risk of Mortality 7.7±6.6%). Technical success was achieved in 96.9% (185/191), and there were no intraprocedural deaths. At 30-day, 1- and 2-year follow-up, the rates of all-cause mortality were 7.9%, 30.8% and 40.5%, respectively. Complete MR elimination (MR <1+) was observed in 99.3%, 99.1% and 96.3% of patients, respectively. TMVR treatment resulted in consistent improvement of New York Heart Association Functional Class and quality of life up to 2 years (both p<0.001). Independent predictors of early mortality were age (odds ratio [OR] 1.11; p=0.003), pulmonary hypertension (OR 3.83; p=0.007), and institutional experience (OR 0.40; p=0.047).
Conclusions: This study investigated clinical outcomes in the full cohort of patients included in the Tendyne Expanded Clinical Study. The Tendyne TMVR system successfully eliminated MR with no intraprocedural deaths, resulting in an improvement in symptoms and quality of life. Continued refinement of clinical and echocardiographic risks will be important to optimise longitudinal outcomes.
Conflict of interest statement
L. Conradi has been an advisory board member for Abbott, Medtronic and JenaValve; and has received personal fees from Boston Scientific, Edwards Lifesciences, Micro Interventional Devices, MicroPort, Neovasc, Pi-Cardia, and Venus Medtech. S. Ludwig has received a grant from the German Heart Foundation (DHS); speaker honoraria from Abbott; travel compensation from Edwards Lifesciences; advisory fees from Bayer; and is a consultant to NVT. D.W. Muller has served as a consultant for Medtronic, Abbott, and Edwards Lifesciences; and has received research grant support from Abbott and Medtronic. P. Sorajja has served as a consultant for 4C Medical, Abbott, Adona, Boston Scientific, Cultiv8, Edwards Lifesciences, Foldax, GE HealthCare, GLG, Laza Medical, Medtronic, Philips, W.L. Gore & Associates, VDyne, and xDot. A. Duncan has served as a proctor and consultant for Abbott, Edwards Lifesciences, Medtronic, and Neochord. B. Bethea has served as a consultant for Abbott. G. Dahle has been proctor/speaker for Abbott; and speaker for Edwards Lifesciences. V. Babaliaros has served as a consultant for Edwards Lifesciences and Abbott; and has equity in Transmural Systems. M. Guerrero has served as a consultant for Edwards Lifesciences and Abbott; and has received research grant support from Edwards Lifesciences. V. Thourani has served as a consultant for and received research grant support from Abbott. N. Dumonteil has been a consultant for Abbott, Boston Scientific, Edwards Lifesciences, and Medtronic. T. Modine has served as a proctor and consultant for Abbott, Edwards Lifesciences, and Medtronic. A. Garatti has served as a proctor for Abbott. P. Denti has received speaker honoraria from Abbott and Edwards Lifesciences; and has been a consultant for Approxima, HVR, InnovHeart, and Pi-Cardia. J. Leipsic has provided institutional core laboratory services to Edwards Lifesciences, Abbott, Medtronic, Boston Scientific, Pi-Cardia, and Neovasc, without direct personal compensation. P. Blanke has provided institutional core laboratory services to Edwards Lifesciences, Abbott, Medtronic, Boston Scientific, Pi-Cardia, and Neovasc, without direct personal compensation; and has been a consultant to Edwards Lifesciences. V. Badhwar has served as an uncompensated consultant for Abbott. M.L. Chuang has no conflicts of interest to declare relevant to the contents of this paper.
Figures

References
-
- Writing Committee Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP 3rd, Gentile F, Jneid H, Krieger EV, Mack M, McLeod C, O’Gara PT, Rigolin VH, Sundt TM 3rd, Thompson A, Toly C. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2021;77:e25–197. - PubMed
-
- Vahanian A, Beyersdorf F, Praz F, Milojevic M, Baldus S, Bauersachs J, Capodanno D, Conradi L, De Bonis, De Paulis, Delgado V, Freemantle N, Haugaa KH, Jeppsson A, Jüni P, Pierard L, Prendergast BD, Sádaba JR, Tribouilloy C, Wojakowski W. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. EuroIntervention. 2022;17:e1126–96. - PMC - PubMed
-
- Mack M, Carroll JD, Thourani V, Vemulapalli S, Squiers J, Manandhar P, Deeb GM, Batchelor W, Herrmann HC, Cohen DJ, Hanzel G, Gleason T, Kirtane A, Desai N, Guibone K, Hardy K, Michaels J, DiMaio JM, Christensen B, Fitzgerald S, Krohn C, Brindis RG, Masoudi F, Bavaria J. Transcatheter Mitral Valve Therapy in the United States: A Report From the STS-ACC TVT Registry. J Am Coll Cardiol. 2021;78:2326–53. - PubMed
-
- Goel SS, Bajaj N, Aggarwal B, Gupta S, Poddar KL, Ige M, Bdair H, Anabtawi A, Rahim S, Whitlow PL, Tuzcu EM, Griffin BP, Stewart WJ, Gillinov M, Blackstone EH, Smedira NG, Oliveira GH, Barzilai B, Menon V, Kapadia SR. Prevalence and outcomes of unoperated patients with severe symptomatic mitral regurgitation and heart failure: comprehensive analysis to determine the potential role of MitraClip for this unmet need. J Am Coll Cardiol. 2014;63:185–6. - PubMed
-
- Ludwig S, Sedighian R, Weimann J, Koell B, Waldschmidt L, Schäfer A, Seiffert M, Westermann D, Reichenspurner H, Blankenberg S, Schofer N, Lubos E, Conradi L, Kalbacher D. Management of patients with mitral regurgitation ineligible for standard therapy undergoing TMVI screening. EuroIntervention. 2022;18:213–23. - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

