Safety and efficacy of PCSK9 inhibitor (evolocumab) in patients with non-ST segment elevation acute coronary syndrome and non-culprit artery critical lesions: a randomised controlled trial protocol (SPECIAL study)
- PMID: 39009458
- PMCID: PMC11253731
- DOI: 10.1136/bmjopen-2023-083730
Safety and efficacy of PCSK9 inhibitor (evolocumab) in patients with non-ST segment elevation acute coronary syndrome and non-culprit artery critical lesions: a randomised controlled trial protocol (SPECIAL study)
Abstract
Introduction: Patients with non-ST segment elevation acute coronary syndrome (NSTE-ACS) and concomitant multivessel coronary artery disease (CAD) are considered patients with extremely high-risk atherosclerotic cardiovascular disease (ASCVD), and current guidelines specify a lower low-density lipoprotein cholesterol (LDL-C) target for this population. Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors have been shown to effectively reduce LDL-C levels on a statin background. Additionally, several studies have confirmed the role of PCSK9 inhibitors in plaque regression and reducing residual cardiovascular risk in patients with ACS. However, those studies included coronary lesions with a degree of stenosis <50%. Whether the application of PCSK9 inhibitors in patients with NSTE-ACS with non-culprit artery critical lesions (stenosis degree between 50% and 75%) has a similar effect on plaque regression and improvement of cardiovascular outcomes remains unknown, with a lack of relevant research. This study aims to further investigate the safety and efficacy of evolocumab in patients with NSTE-ACS and concomitant multivessel CAD (non-culprit artery stenosis between 50% and 75%).
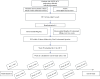
Methods and analysis: In this single-centre clinical randomised controlled trial, 122 patients with NSTE-ACS and concomitant multivessel CAD (non-culprit artery stenosis between 50% and 75%) will be randomly assigned to either the evolocumab treatment group or the standard treatment group after completing culprit vessel revascularisation. The evolocumab treatment group will receive evolocumab in addition to statin therapy, while the standard treatment group will receive standard statin therapy. At baseline and week 50, patients in the evolocumab treatment group will undergo coronary angiography and OCT imaging to visualise pre-existing non-lesional vessels. The primary end point is the absolute change in average minimum fibrous cap thickness (FCT) from baseline to week 50. Secondary end points include changes in plaque lipid arc, lipid length, macrophage grading, lipid levels and major adverse cardiovascular events during the 1-year follow-up period.
Ethics and dissemination: Ethics: this study will adhere to the principles outlined in the Helsinki Declaration and other applicable ethical guidelines. This study protocol has received approval from the Medical Research Ethics Committee of the First Affiliated Hospital of the University of Science and Technology of China (Anhui Provincial Hospital), with approval number 2022-ky214.
Dissemination: we plan to disseminate the findings of this study through various channels. This includes publication in peer-reviewed academic journals, presentation at relevant academic conferences and communication to the public, policymakers and healthcare professionals. We will also share updates on the research progress through social media and other online platforms to facilitate the exchange and application of scientific knowledge. Efforts will be made to ensure widespread dissemination of the research results and to have a positive impact on society.
Trial registration number: ChiCTR2200066675.
Keywords: cardiovascular disease; coronary heart disease; coronary intervention; myocardial infarction.
© Author(s) (or their employer(s)) 2024. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures
References
-
- Steg PG, Goldberg RJ, Gore JM, et al. Baseline characteristics, management practices, and in-hospital outcomes of patients hospitalized with acute coronary syndromes in the global Registry of acute coronary events (GRACE) Am J Cardiol. 2002;90:358–63. doi: 10.1016/s0002-9149(02)02489-x. - DOI - PubMed
-
- Sato A, Ohigashi H, Nozato T, et al. Coronary artery spatial distribution, morphology, and composition of Nonculprit coronary plaques by 64-slice computed Tomographic angiography in patients with acute myocardial infarction. Am J Cardiol. 2010;105:930–5. doi: 10.1016/j.amjcard.2009.11.028. - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous