AI-Powered Mining of Highly Customized and Superior ESIPT-Based Fluorescent Probes
- PMID: 39021325
- PMCID: PMC11425259
- DOI: 10.1002/advs.202405596
AI-Powered Mining of Highly Customized and Superior ESIPT-Based Fluorescent Probes
Abstract
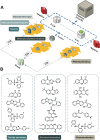
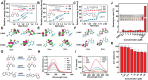
Excited-state intramolecular proton transfer (ESIPT) has attracted great attention in fluorescent sensors and luminescent materials due to its unique photobiological and photochemical features. However, the current structures are far from meeting the specific demands for ESIPT molecules in different scenarios; the try-and-error development method is labor-intensive and costly. Therefore, it is imperative to devise novel approaches for the exploration of promising ESIPT fluorophores. This research proposes an artificial intelligence approach aiming at exploring ESIPT molecules efficiently. The first high-quality ESIPT dataset and a multi-level prediction system are constructed that realized accurate identification of ESIPT molecules from a large number of compounds under a stepwise distinguishing from conventional molecules to fluorescent molecules and then to ESIPT molecules. Furthermore, key structural features that contributed to ESIPT are revealed by using the SHapley Additive exPlanations (SHAP) method. Then three strategies are proposed to ensure the ESIPT process while keeping good safety, pharmacokinetic properties, and novel structures. With these strategies, >700 previously unreported ESIPT molecules are screened from a large pool of 570 000 compounds. The ESIPT process and biosafety of optimal molecules are successfully validated by quantitative calculation and experiment. This novel approach is expected to bring a new paradigm for exploring ideal ESIPT molecules.
Keywords: ESIPT; artificial intelligence; fluorescent probe; machine learning; virtual screening.
© 2024 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Sedgwick A. C., Dou W. T., Jiao J. B., Wu L., Williams G. T., Jenkins A. T. A., Bull S. D., Sessler J. L., He X. P., James T. D., J. Am. Chem. Soc. 2018, 140, 14267. - PubMed
-
- Hu R., Feng J., Hu D., Wang S., Li S., Li Y., Yang G., Angew. Chem., Int. Ed. Engl. 2010, 49, 4915. - PubMed
-
- Fu P.‐Y., Yi S.‐Z., Pan M., Su C.‐Y., Acc. Mater. Res. 2023, 4, 939.
-
- Huang Q., Guo Q., Lan J., Su R., Ran Y., Yang Y., Bin Z., You J., Mater. Horiz. 2021, 8, 1499. - PubMed
-
- Hsieh C.‐C., Jiang C.‐M., Chou P.‐T., Acc. Chem. Res. 2010, 43, 1364. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
