Bone marrow-derived myeloid cells transiently colonize the brain during postnatal development and interact with glutamatergic synapses
- PMID: 39021809
- PMCID: PMC11253522
- DOI: 10.1016/j.isci.2024.110037
Bone marrow-derived myeloid cells transiently colonize the brain during postnatal development and interact with glutamatergic synapses
Abstract
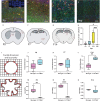
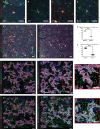
Although the roles of embryonic yolk sac-derived, resident microglia in neurodevelopment were extensively studied, the possible involvement of bone marrow-derived cells remains elusive. In this work, we used a fate-mapping strategy to selectively label bone marrow-derived cells and their progeny in the brain (FLT3+IBA1+). FLT3+IBA1+ cells were confirmed to be transiently present in the healthy brain during early postnatal development. FLT3+IBA1+ cells have a distinct morphology index at postnatal day(P)0, P7, and P14 compared with neighboring microglia. FLT3+IBA1+ cells also express the microglial markers P2RY12 and TMEM119 and interact with VGLUT1 synapses at P14. Scanning electron microscopy indeed showed that FLT3+ cells contact and engulf pre-synaptic elements. Our findings suggest FLT3+IBA1+ cells might assist microglia in their physiological functions in the developing brain including synaptic pruning which is performed using their purinergic sensors. Our findings stimulate further investigation on the involvement of peripheral macrophages during homeostatic and pathological development.
Keywords: Biological sciences; Cellular neuroscience; Developmental neuroscience; Natural sciences; Neuroscience.
© 2024 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Yona S., Kim K.-W., Wolf Y., Mildner A., Varol D., Breker M., Strauss-Ayali D., Viukov S., Guilliams M., Misharin A., et al. Fate Mapping Reveals Origins and Dynamics of Monocytes and Tissue Macrophages under Homeostasis. Immunity. 2013;38:79–91. doi: 10.1016/j.immuni.2012.12.001. - DOI - PMC - PubMed
-
- Cecchini M.G., Dominguez M.G., Mocci S., Wetterwald A., Felix R., Fleisch H., Chisholm O., Hofstetter W., Pollard J.W., Stanley E.R. Role of colony stimulating factor-1 in the establishment and regulation of tissue macrophages during postnatal development of the mouse. Development. 1994;120:1357–1372. - PubMed
-
- Hu H., Yang X., He Y., Duan C., Sun N. Psychological stress induces depressive-like behavior associated with bone marrow-derived monocyte infiltration into the hippocampus independent of blood–brain barrier disruption. J. Neuroinflammation. 2022;19:208. doi: 10.1186/s12974-022-02569-w. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

