Food allergy oral immunotherapy
- PMID: 39022134
- PMCID: PMC11250211
- DOI: 10.2500/jfa.2020.2.200005
Food allergy oral immunotherapy
Abstract
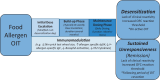
Food allergy oral immunotherapy (OIT) has demonstrated efficacy in promoting clinically relevant immunomodulation that leads to desensitization (reduced reactivity while on OIT) in the majority of treated individuals; however, sustained unresponsiveness after OIT cessation for a specified interval has only been observed in a subset. The potential therapeutic benefits of OIT must be balanced with the risk for adverse events. These adverse events may range from self-limited or easily treated oropharyngeal, respiratory, or gastrointestinal symptoms to persistent abdominal symptoms that lead to cessation of therapy and to anaphylaxis. To date, the majority of studies have evaluated single-allergen OIT approaches; however, multi-allergen OIT has demonstrated favorable safety and efficacy outcomes, and is the subject of ongoing investigation. Recent U.S. Food and Drug Administration approval of the first licensed OIT product for peanut allergy challenges the long-standing paradigm of dietary food avoidance as the sole option for individuals with food allergy. Yet, the limitations of this "first-generation" treatment support the need for continued research and development of next-generation therapies to improve efficacy, minimize risk, and allow for broad applicability to both individuals with single-food allergy and those with multifood allergies. Optimizing future therapies will require developing novel approaches that maximize both efficacy and safety and/or tolerability outcomes, potentially through the combination with biologic therapies or adjuvants. Shared decision-making among patients, physicians, and parents and/or caregivers is critical to select optimal candidates for treatment with OIT by balancing the potential therapeutic benefit and possible risk reduction with a realistic consideration of OIT treatment burden and the risk of treatment-related adverse events.
Copyright © 2020, The Author(s). Published by OceanSide Publications, Inc., U.S.A.
Conflict of interest statement
The authors have no conflicts of interest to declare pertaining to this article
Figures

References
LinkOut - more resources
Full Text Sources
