Molecular fingerprints of cardiovascular toxicities of immune checkpoint inhibitors
- PMID: 39023770
- PMCID: PMC11790702
- DOI: 10.1007/s00395-024-01068-8
Molecular fingerprints of cardiovascular toxicities of immune checkpoint inhibitors
Abstract
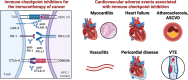
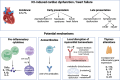
Immune checkpoint inhibitors (ICIs) have revolutionized cancer therapy by unleashing the power of the immune system against malignant cells. However, their use is associated with a spectrum of adverse effects, including cardiovascular complications, which can pose significant clinical challenges. Several mechanisms contribute to cardiovascular toxicity associated with ICIs. First, the dysregulation of immune checkpoints, such as cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein-1 (PD-1) and its ligand (PD-L1), and molecular mimicry with cardiac autoantigens, leads to immune-related adverse events, including myocarditis and vasculitis. These events result from the aberrant activation of T cells against self-antigens within the myocardium or vascular endothelium. Second, the disruption of immune homeostasis by ICIs can lead to autoimmune-mediated inflammation of cardiac tissues, manifesting as cardiac dysfunction and heart failure, arrhythmias, or pericarditis. Furthermore, the upregulation of inflammatory cytokines, particularly tumor necrosis factor-alpha, interferon-γ, interleukin-1β, interleukin-6, and interleukin-17 contributes to cardiac and endothelial dysfunction, plaque destabilization, and thrombosis, exacerbating cardiovascular risk on the long term. Understanding the intricate mechanisms of cardiovascular side effects induced by ICIs is crucial for optimizing patient care and to ensure the safe and effective integration of immunotherapy into a broader range of cancer treatment protocols. The clinical implications of these mechanisms underscore the importance of vigilant monitoring and early detection of cardiovascular toxicity in patients receiving ICIs. Future use of these key pathological mediators as biomarkers may aid in prompt diagnosis of cardiotoxicity and will allow timely interventions.
Keywords: Autoimmune; Cardiotoxicity; Immune-related adverse event; Immunotherapy.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: The authors declare no conflict of interest. Dr. Ferdinandy is the founder and CEO of Pharmahungary Group, a group of R&D companies.
Figures



Similar articles
-
Mechanisms and clinical manifestations of cardiovascular toxicities associated with immune checkpoint inhibitors.Clin Sci (Lond). 2021 Mar 12;135(5):703-724. doi: 10.1042/CS20200331. Clin Sci (Lond). 2021. PMID: 33686402 Free PMC article. Review.
-
Cardiovascular toxicities associated with immune checkpoint inhibitors.Cardiovasc Res. 2019 Apr 15;115(5):854-868. doi: 10.1093/cvr/cvz026. Cardiovasc Res. 2019. PMID: 30715219 Free PMC article. Review.
-
How can we manage the cardiac toxicity of immune checkpoint inhibitors?Expert Opin Drug Saf. 2021 Jun;20(6):685-694. doi: 10.1080/14740338.2021.1906860. Epub 2021 Apr 1. Expert Opin Drug Saf. 2021. PMID: 33749484 Review.
-
Immune checkpoint inhibitors associated cardiovascular immune-related adverse events.Front Immunol. 2024 Feb 5;15:1340373. doi: 10.3389/fimmu.2024.1340373. eCollection 2024. Front Immunol. 2024. PMID: 38375475 Free PMC article. Review.
-
Insight of immune checkpoint inhibitor related myocarditis.Int Immunopharmacol. 2024 Dec 25;143(Pt 3):113559. doi: 10.1016/j.intimp.2024.113559. Epub 2024 Nov 12. Int Immunopharmacol. 2024. PMID: 39536487 Review.
Cited by
-
AI-Assisted Hypothesis Generation to Address Challenges in Cardiotoxicity Research: Simulation Study Using ChatGPT With GPT-4o.J Med Internet Res. 2025 May 15;27:e66161. doi: 10.2196/66161. J Med Internet Res. 2025. PMID: 40373295 Free PMC article.
-
Immuno-related cardio-vascular adverse events associated with immuno-oncological treatments: an under-estimated threat for cancer patients.Basic Res Cardiol. 2025 Feb;120(1):153-169. doi: 10.1007/s00395-024-01077-7. Epub 2024 Sep 3. Basic Res Cardiol. 2025. PMID: 39225869 Free PMC article. Review.
-
The cardio-oncologic burden of breast cancer: molecular mechanisms and importance of preclinical models.Basic Res Cardiol. 2025 Feb;120(1):91-112. doi: 10.1007/s00395-024-01090-w. Epub 2024 Dec 2. Basic Res Cardiol. 2025. PMID: 39621070 Free PMC article. Review.
-
Cryptogenic embolic stroke and cancer.Front Neurol. 2025 Jun 13;16:1537779. doi: 10.3389/fneur.2025.1537779. eCollection 2025. Front Neurol. 2025. PMID: 40584524 Free PMC article. Review.
-
Immune checkpoint inhibitor-induced thyroiditis and its potential mechanisms.Front Endocrinol (Lausanne). 2025 Jun 4;16:1584675. doi: 10.3389/fendo.2025.1584675. eCollection 2025. Front Endocrinol (Lausanne). 2025. PMID: 40535343 Free PMC article. Review.
References
-
- Advani R, Flinn I, Popplewell L, Forero A, Bartlett NL, Ghosh N, Kline J, Roschewski M, LaCasce A, Collins GP, Tran T, Lynn J, Chen JY, Volkmer J-P, Agoram B, Huang J, Majeti R, Weissman IL, Takimoto CH, Chao MP, Smith SM (2018) CD47 blockade by Hu5F9-G4 and rituximab in non-hodgkin’s lymphoma. N Engl J Med 379:1711–1721. 10.1056/nejmoa1807315 - PMC - PubMed
-
- Ait-Oufella H, Salomon BL, Potteaux S, Robertson AKL, Gourdy P, Zoll J, Merval R, Esposito B, Cohen JL, Fisson S, Flavell RA, Hansson GK, Klatzmann D, Tedgui A, Mallat Z (2006) Natural regulatory T cells control the development of atherosclerosis in mice. Nat Med 12:178–180. 10.1038/nm1343 - PubMed
-
- Alhumaid W, Yogasundaram H, Senaratne JM (2021) Slow bidirectional ventricular tachycardia as a manifestation of immune checkpoint inhibitor myocarditis. Eur Heart J 42:2868. 10.1093/eurheartj/ehab219 - PubMed
-
- Allenbach Y, Anquetil C, Manouchehri A, Benveniste O, Lambotte O, Lebrun-Vignes B, Spano JP, Ederhy S, Klatzmann D, Rosenzwajg M, Fautrel B, Cadranel J, Johnson DB, Moslehi JJ, Salem JE (2020) Immune checkpoint inhibitor-induced myositis, the earliest and most lethal complication among rheumatic and musculoskeletal toxicities. Autoimmun Rev 19:102586. 10.1016/j.autrev.2020.102586 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

